- NEWS AND VIEWS
A two-pronged strategy directs drug-delivering nanoparticles to the pancreas — and shows promise in animal models of serious pancreatic diseases.
By
- Aviad Elisha
-
Aviad Elisha is at the Shmunis School of Biomedicine and Cancer Research, Tel Aviv University, Tel Aviv, Israel, and in the Department of Materials Sciences and Engineering, the Center for Nanoscience and Nanotechnology and the Cancer Biology Research Center, Tel Aviv University.
-
- Dan Peer
-
Dan Peer is at the Shmunis School of Biomedicine and Cancer Research, Tel Aviv University, Tel Aviv, Israel, and in the Department of Materials Sciences and Engineering, the Center for Nanoscience and Nanotechnology and the Cancer Biology Research Center, Tel Aviv University.
-
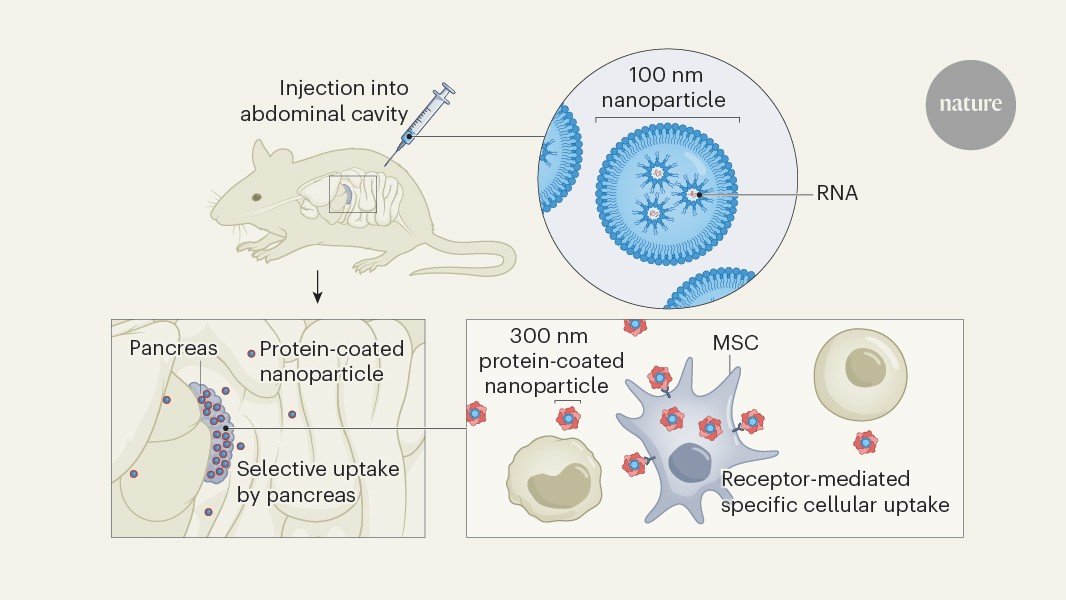
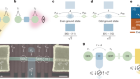
The clinical use of lipid nanoparticles (LNPs) for drug delivery is currently limited by their tendency to accumulate in the liver. Tuning their biodistribution to reach other tissues and to target specific cell types in an organ remains a major challenge1,2. Writing in Nature, Lei et al.3 report their approach for engineering LNPs that specifically deliver therapeutic RNA to the pancreas — successfully targeting cell populations that were previously beyond the reach of LNPs, and unlocking avenues of research towards therapies that combat currently incurable pancreatic diseases.
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
doi: https://doi.org/10.1038/d41586-026-00294-5
References
-
Peer, D. et al. Nature Nanotechnol. 2, 751–760 (2007).
-
Kon, E., Ad-El, N., Hazan-Halevy, I., Stotsky-Oterin, L. & Peer, D. Nature Rev. Clin. Oncol. 20, 739–754 (2023).
-
Lei, J. et al. Nature https://doi.org/10.1038/s41586-026-10158-7 (2026).
-
Wang, X. et al. Nature Protoc. 18, 265–291 (2023).
-
Chan, W. C. W. BME Front. 4, 0016 (2023).
-
Hamilton, A. G. et al. ACS Nano. 18, 16151–16165 (2024).
-
Rampado, R. et al. Adv. Sci. 12, e2408744 (2025).
-
Tarab-Ravski, D. et al. J. Control Release 376, 286–302 (2024).
-
Lam, K. et al. Adv. Mater. 35, e2211420 (2023).
Competing Interests
D. P. receives unrelated licensing fees (to patents on which he was an inventor) from, has invested in, consults for (or is on scientific advisory boards or boards of directors for), has lectured for (and received a fee) and conducts sponsored research at Tel Aviv University for: ART Biosciences; BioNtech SE; Earli Inc.; Kernal Biologics; LAND Therapeutics; Merck KGaA; Newphase Ltd.; NeoVac Ltd.; RiboX Therapeutics; Roche; SirTLabs Corporation; Teva Pharmaceuticals Inc.
Related Articles
-
 Read the paper: Pancreatic-targeted lipid nanoparticles based on organ capsule filtration
Read the paper: Pancreatic-targeted lipid nanoparticles based on organ capsule filtration -
 Tiny robots swim through blood, deliver drugs — and then dissolve
Tiny robots swim through blood, deliver drugs — and then dissolve -
 Stop the nonsense: genome editing creates potentially therapeutic transfer RNAs
Stop the nonsense: genome editing creates potentially therapeutic transfer RNAs