The value of ex vivo autologous CAR T therapies is clear: for some patients, these treatments offer a second chance at life. Yet the approach comes with significant limitations. The patient-physician journey is complex, and manufacturing is both costly and logistically demanding, due to reliance on ex vivo manipulation of each patient’s apheresis-derived T cells under GMP conditions.
The number of patients treated represents only a small fraction of those in need, limiting democratization of this groundbreaking treatment. Will in vivo CAR T therapies, positioned as off-the-shelf products, be able to break down these barriers and expand access?
A range of in vivo CAR T platforms—both integrated and non-integrated—are currently under investigation. Integrated approaches typically rely on viral vectors, such as lentivirus, similar to those used in most approved CAR T therapies. As with other viral gene therapy strategies, these platforms carry known risks, including the potential for insertional mutagenesis.
Despite these risks, early clinical data suggest meaningful potential for in vivo CAR T approaches. Two early studies—one performed by Belgium-based EsoBiotec in collaboration with China-based Shenzhen Pregene Biopharma, and another by Boston-based Kelonia Therapeutics—have demonstrated tumor clearance in multiple myeloma using lentiviral vectors.
Complementary non-integrated platforms use a variety of delivery methods, including lipid nanoparticles (LNPs), nanosomes, and exosomes. In these scenarios, the half-life of mRNA, the primary delivery vehicle, is a challenge, likely requiring the ability to redose without immunogenicity. DNA-based payloads are being investigated as alternatives.
Major pharmaceutical companies are racing to acquire in vivo cell engineering platforms, signaling that reprogramming cells inside the body is becoming a central pillar of therapeutic strategy. A wave of company consolidations demonstrates the impending expansion of this nascent, yet rapidly maturing, field beyond oncology applications to other disease arenas such as autoimmune disorders.
Specificity and scaling challenges
Early in 2021, Sanofi saw the potential and purchased Tidal Therapeutics. At the time, Sanofi anticipated that this next-generation, off-the-shelf approach had the potential to bring CAR T cell therapy to a much broader patient population.
“To increase efficiency, we are working on non-integrated approaches that are targeted to deliver mRNA cargoes to specific cell types,” said Christian Mueller, PhD, vice president and global head of genomic medicine at Sanofi. Sanofi’s CD8-targeted LNP efficiently delivers CD19 CAR mRNA to CD8 T cells.
The ionizable lipid and its formulation enable efficient transfection of T cells in vitro and in animal models. In nonhuman primate (NHP) studies, administration of the targeted LNPs resulted in profound and sustained B cell depletion with few, and mostly transient, subclinical side effects.
“We engineer our targeting moieties to ensure that we have site-specific lipidation for incorporation into the LNP,” explained Mueller. The site-specific location of the modification ensures batch-to-batch consistency and robust scalability relative to other LNP targeting methodologies.
Scaling can be challenging. Targeted LNPs require the manufacture of several different components, such as mRNA, lipids, and targeting moieties, all of which need to be formulated into a consistent drug product. Significant engineering development at Sanofi has enabled an efficient targeted LNP production process.
Mueller predicts the durability of in vivo CAR expression will continue to improve. Multiple strategies exist, including the potential for LNP-mediated delivery of DNA to enable longer dosing windows, or even permanent expression of a CAR through gene insertion technologies. Continued development work over the next few years will reveal the appropriateness of these advanced payloads for varied applications.
CARs and CAR targets will continue to diversify beyond hematologic applications. Engineering may permit more complex circuitry that could unlock the full potential of an in vivo cell reprogramming approach, including, but not limited to, expression of factors to allow for the treatment of solid tumors.
Sanofi is considering extended applications of their targeted LNP platform beyond in vivo CAR T to reprogramming of other cell types. In particular, the targeting of hematopoietic stem cells is interesting and could deliver potentially curative therapies to patients.
Acquisitions ramp up
More recently, industry dealmaking has accelerated. According to Oncology Pipeline, in 2025, Bristol Myers Squibb (BMS) acquired Orbital Therapeutics and its RNA-based in vivo pipeline for $1.5 billion. Gilead, through its subsidiary Kite, committed $120 million up front and up to $1.5 billion in milestone payments to partner with Chinese cell therapy specialist Pregene, before subsequently acquiring Interius BioTherapeutics.

Other large pharmaceutical companies have made similar moves. AstraZeneca acquired EsoBiotec for up to $1 billion, while AbbVie made headlines with its $2.1 billion acquisition of Capstan Therapeutics and a separate licensing agreement with Umoja. Kelonia Therapeutics has also announced a strategic collaboration with Johnson & Johnson to advance the discovery of novel in vivo CAR T-cell therapies.
Leveraging platform experience
The pace of technology and platform development in the 2020s far exceeds that of previous decades. AI-enabled access to information strengthens hypothesis generation while evidence-based knowledge accumulation from global trial networks accelerates clinical development.
“Although in vivo CAR generation is a nascent field, we have signals of great potential. The work done on ex vivo therapies, including Yescarta® and Tecartus®, that have been used to treat almost 23,000 patients to date, provides confidence in the approach with prior knowledge for comparison,” said Priti Hegde, PhD, senior vice president and global head of research, Kite, a Gilead Company. “The level of investment is significant. The applications are vast. If we can get these platforms to work, it will be game changing.”
An outstanding question is whether a CAR alone has the ability to do its job, expand, and kill target cells in the absence of a lymphodepletion regimen to eliminate immunosuppressant cells. “Clinical data and long-term patient monitoring are needed to understand if in vivo approaches are as durable and provide similar clinical characteristics as ex vivo platforms,” emphasized Hegde.
Due to experience with lentiviral ex vivo platforms, Kite elected to use a lentivirus-based platform for in vivo CAR development work. To prevent non-specific delivery, the engineered virus is coated with a targeting moiety to specifically deliver the payload to a cell expressing the target. Changing the targeting binder and the payload provides diversity for application to different cell types and diseases, as well as for crucial IP creation.
Hegde pointed out the critical importance of manufacturing at scale. “We have invested a lot of effort into manufacturing lentivirus. The acquisition of Interius BioTherapeutics complements our internal development work.”
Interius’s scalable modular model for viral vectors allows faster program development. The first program in dose escalation Phase I studies in Australia and Europe is targeted towards CD20 in lymphomas and uses a non-activating binder that targets CD7 on T cells. “Now that the manufacturing program is integrated into Kite Research, we are exploring other binders and CAR constructs,” Hegde added.
Taking both approaches
AbbVie is currently investigating in vivo CAR T platforms across immune-mediated diseases and cancer.
“In vivo CAR T cell approaches could transform how we address difficult-to-treat autoimmune diseases and cancers, potentially bringing us closer to the possibility of true cures,” said Jonathon Sedgwick, PhD, senior vice president and global head of discovery research at AbbVie. “The approach could overcome major hurdles of autologous CAR T therapies and may eventually help expand the patient population benefiting from CAR T therapies.”
One method uses a proprietary targeted LNP (tLNP) platform for RNA delivery, initially developed by Capstan Therapeutics. The tLNPs are designed to minimize off-target effects, including liver de-targeting chemistry. Targeting binders on the LNPs that recognize specific T cell surface markers enables delivery of RNA to desired T cell populations, transiently converting them into CAR-expressing cells that can seek and destroy pathogenic cells known to drive certain autoimmune diseases.
According to Sedgwick, ABBV-619 is designed to program CD8+ T cells in the patient’s body to express CD19-specific CARs. By generating these CD19 CAR-expressing cytotoxic CD8 T cells, the approach aims to deplete pathogenic memory B cells in the periphery and tissues that, in turn, enables repopulation with naïve B cells, potentially resetting the immune system to halt or reverse disease progression. ABBV-619 is currently in Phase I studies for the treatment of B cell-mediated autoimmune diseases.
The company’s partnership with Umoja leverages the proprietary VivoVec platform to develop in vivo CAR T therapies for oncology. The platform combines third-generation lentiviral vector gene delivery with a novel T cell targeting and activation surface complex. VivoVec can be combined with RACR (rapamycin-activated cytokine receptor) to potentially enhance survival and expansion of VivoVec-engineered immune cells.
platform to develop in vivo CAR T therapies for oncology. The platform combines third-generation lentiviral vector gene delivery with a novel T cell targeting and activation surface complex. VivoVec can be combined with RACR (rapamycin-activated cytokine receptor) to potentially enhance survival and expansion of VivoVec-engineered immune cells.
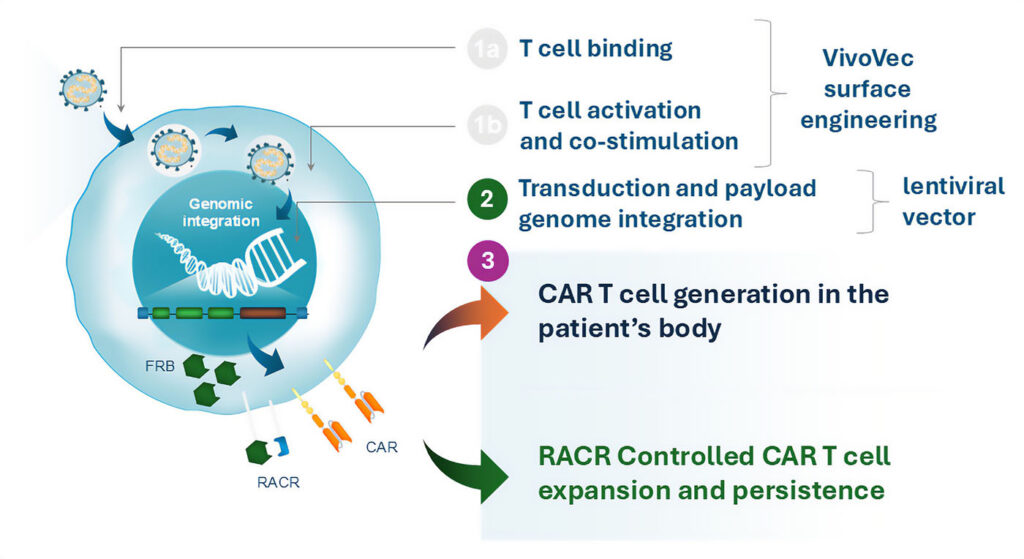
AbbVie holds an exclusive option to license Umoja’s CD19-directed in vivo CAR T candidates, including UB-VV111, Umoja’s lead clinical program for hematological malignancies currently in Phase I studies.
“By drawing upon our deep heritage in immunology and oncology, leveraging our internal capabilities, along with strategic investments and partnerships, we are committed to advancing in vivo CAR T platforms to create more accessible, durable, and transformative therapies to target a broad range of pathogenic cell types,” said Sedgwick.
The post In Vivo CAR T Therapy Will the Bets Pay Off? appeared first on GEN – Genetic Engineering and Biotechnology News.