Introduction
Organoids, derived from among others induced pluripotent stem cells (iPSCs), are self-organizing 3D-multicellular structures that mimic organ functions in vitro and have become a popular source for studying organ development, drug discovery, and various diseases1. Compared to conventional 2D cultures and animal models, organoid culture allows patient specificity while recapitulating in vivo tissue-like structures and functions in vitro. Specifically, BVOs or vascular organoids, also derived from hiPSCs are favored in vascular regeneration research. Vascular organoids were first reported in 2019 and are composed of organized endothelial cells (ECs) and mural cells and have demonstrated functionality when implanted under the renal capsule of mice, connecting host vasculature. Vascular organoids are often expanded in extracellular matrix (ECM) protein-based hydrogels, with Matrigel being the most common choice2,3. Matrigel, first described in the 1980s, is a reconstituted basement-membrane matrix derived from Engelbreth-Holm-Swarm mouse sarcomas, which is rich in laminin, collagen IV and other ECM proteins4,5,6. The high biocompatibility of Matrigel makes it a popular substrate for hiPSCs as well as embryonic stem cells (ESCs), enabling cell adhesion, proliferation and differentiation while providing essential cytokines for chemical signaling, along with ECM proteins for structural support and necessary to replicate the natural 3D in vivo environment. While Matrigel promotes vascular organoid maturation and angiogenic sprouting, its high batch-to-batch differences in composition lead to difficulties in reproducibility. Additionally, due to its murine origin and presence of tumor-derived growth factors it cannot be used for translational therapies4. Therefore, there is a need to find an animal-free hydrogel that can support vascular organoid growth and can be translated into a human setting. Additionally, the use of animal-free, human-derived materials is highly relevant for regenerative medicine applications, including drug screening, the development of novel therapeutics targeting vascular diseases, and the creation of physiologically relevant inflammatory models. Their human origin eliminates cross-species immune activation and enhances translational relevance to humans.
Matrigel coating is also commonly used for 2D hiPSC culture, however, some animal-free alternatives have been proposed as coating materials. Cell-adhesive ECMs such as Vitronectin, laminin (laminin-511 recombinant laminin, and laminin fragments) are promising approaches to establish the fully defined cell culture conditions for clinical grade hiPSCs7,8,9. However, it has been described that coating influences the growth and expansion of hiPSCs as well as differentiation efficacy10. Additionally, different cell lines may exhibit different responses to substrates, making it essential to test the chosen coating method for the desired application. For this project, the chosen substrate was Vitronectin XF™ (Vitronectin), a xeno-free matrix that supports the growth and differentiation of hiPSCs under serum- and feeder-free conditions. Vitronectin is a recombinant human protein that enables enzyme free passaging, supports mesoderm induction, and facilitates downstream differentiation, making it a technically suitable option for hiPSC-derived vascular organoid culture11,12,13. While much of the research into Matrigel replacement often focuses on 2D cell culture approaches there has been a growing interest in developing suitable animal-free 3D culture conditions including GrowDex® hydrogel, VitroGel® and Manchester BIOGEL, that may be suitable for vascular organoid applications14. However, current literature indicates that these materials have not yet been demonstrated to fully support the generation and maturation of iPSC-derived vascular organoids containing both endothelial and mural cells in a manner equivalent to Matrigel. Recent reviews highlight the promise and ongoing development of synthetic and animal-free hydrogels for organoid culture, but also emphasize that challenges remain in recapitulating the complex microenvironment required for robust co-differentiation and organization of multiple vascular cell types15,16.
For 3D hydrogel replacement of the traditional Matrigel, the alternative should possess similar characteristics in terms of stiffness and polymerization, allowing sprouting behavior. In any method of microvascular network formation, the alignment of the formed micro vessels is critical, as it designates the inlet and outlet sides of the tissue for in vitro perfusion17. Many chemical alternatives used as animal-free culture substrates are not suitable for this application due to limitations in stiffness or a lack of biofunctionalization, such as the presence of ECM bound growth factors.
Previously, Nguyen et al. (2017) identified synthetic poly(ethylene glycol) (PEG) hydrogels as an alternative substrate for in vitro angiogenesis, supporting endothelial sprouting and maintenance in 2D18. While PEG’s stability, hydrophilicity, and protein resistance make it suitable for cell encapsulation19, impurities like ethylene oxide and 1,4-dioxane—carcinogens and respiratory irritants—limit its clinical applicability due to potential neurotoxicity from prolonged exposure.
Fibrin is a widely used 3D matrix for endothelial cell maintenance and sprout formation due to its natural angiogenic properties and role as a provisional matrix in wound healing17,20,21,22. Growth factors within fibrin gels are gradually released, promoting endothelial assembly over several days22. Fibrin polymerizes when thrombin, generated from prothrombin during blood coagulation, acts on fibrinogen, a plasma protein produced in the liver.
In vitro, combining thrombin and fibrinogen forms a fibrin gel suitable for vascular organoid culture. Fibrin-based hydrogels are favored for their biocompatibility, non-toxic degradation, support for cell proliferation, and versatility, with gelation time adjustable via thrombin concentration23,24. These hydrogels have been shown to support epithelial organoids, mesenchymal stem cells (MSCs), endothelial cells, and angiogenesis, making them a promising option for vascular organoid differentiation23,25,26,27.
Compared to other animal-free matrices, the use of Vitronectin- and fibrin-based hydrogels offers a human-derived, recombinant, and xeno-free platform ideal for regenerative medicine. Their defined composition minimizes batch variability and cross-species immune responses, enhancing clinical translatability and safety. These matrices also support reproducible disease modelling, particularly in animal-free 3D vascular organoid systems, and can be engineered to present precise biochemical and mechanical cues that promote the expansion and differentiation of pluripotent stem cells and vascular cell types. Therefore, in this study, we aim to replace Matrigel in vascular organoid culture with animal-free materials to bridge the gap between in vitro models and in vivo applications. Human-derived Vitronectin provides a cost-effective and viable alternative for hiPSC culture prior to differentiation. For 3D vascular organoid culture, fibrinogen and thrombin emerge as promising substitutes due to their biocompatibility, availability, cost-efficiency, and capacity for modification to mimic the cell’s natural environment. Adjusting the ratio of fibrinogen to thrombin allows for control over polymerization speed and gel mechanics, making it adaptable to the vascular organoid’s needs. Our findings demonstrate that Matrigel can be effectively replaced with a Vitronectin-fibrin system, rendering the vascular organoid differentiation protocol entirely animal-free and in support of its clinical translation.
Results
The hiPSC lines used in this study are; SCV1273 (referred to as Clone A), UKKi032-C (NP0141-31B, referred to as Clone 1), and UKKi036-C (NP0143-18, referred to as Clone 2).
In this study, 2 different hiPSC coatings and 3 different hydrogel regimes were evaluated, displayed in Table 1.
HiPSCs cultured on Vitronectin-coated substrate yield similar pluripotent characteristics
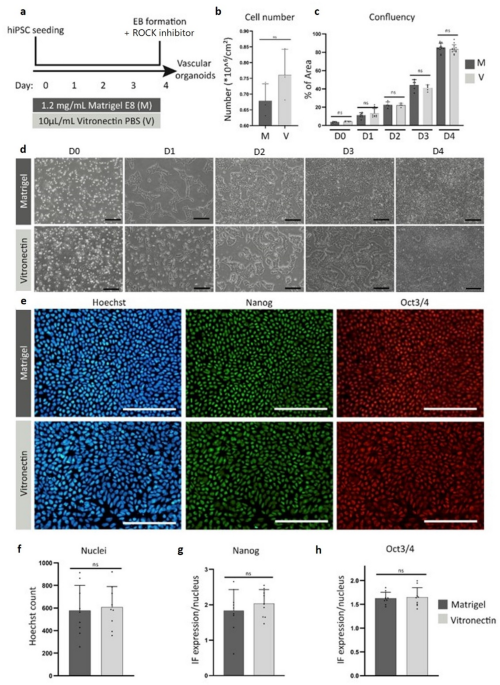
hiPSCs were cultured on either Matrigel or Vitronectin for 5 consecutive days before passaging (Fig. 1a). Based on cell number, confluency, and morphology, no significant differences were detected between the two groups (Fig. 1b-d). Cells were stained for pluripotency markers Nanog and OCT3/4 and counterstained with Hoechst (Fig. 1e), and quantification revealed no significant differences (Fig. 1f-h). iPSC clones 1 and 2 demonstrate similar results compared to clone A (Supplementary Fig. S1). These results indicate that Vitronectin is a good animal-free substitute for Matrigel.
hiPSCs cultured on Vitronectin-coated substrate yield similar pluripotent characteristics. Data shown are from Clone A. (a) Schematic timeline of culturing hiPSCs to embryoid bodies in different coating conditions. (b) Total cell number during 4 days of culturing. Error bars represent SD (standard deviation) of n = 3 experiments for both groups. (c) Confluency analysis per day of culturing. Error bars represent SD of n = 4 experiments for both groups, for each n, 3 technical replicates (3 wells). (d) hiPSCs cultured on Matrigel or Vitronectin during one passage (day 0–4). Scale bar depicts 200 μm. (e) Immunostaining of hiPSC cultured on Matrigel or Vitronectin on day 4. Cells were stained for pluripotency markers NANOG, OCT3/4, and nuclei counterstained with Hoechst (left panel). Scale bars depict 200 μm. (f) Total nuclei amount on day 4 of culturing. Error bars represent SD of n = 3 experiments for both groups, for each n, 3 technical replicates. (g) Quantification of pluripotency marker Nanog on day 4 of culturing. Error bars represent SD of n = 3 experiments for both groups, for each n, 3 technical replicates. (h) Quantification of pluripotency marker OCT3/4 on day 4 of culturing. Error bars represent SD of n = 3 experiments for both groups, for each n, 3 technical replicates. Significance is determined by unpaired T-test (b, f-h) or One-way ANOVA (c), and no significant differences between the iPSC groups were detected.
Vascular organoids derived from Vitronectin-based hiPSCs show a similar differentiation pattern compared to Matrigel based hiPSCs
Vascular organoid differentiation takes 18–21 days, involving two Matrigel-based steps, 2D iPSC culture (day 0) and 3D vascular organoid differentiation (days 13–21+) (Fig. 2a). To determine whether the use of Vitronectin coating for hiPSC culture impairs differentiation, the differentiation protocol was followed as described in previous research27,29. During the differentiation process, brightfield images were taken to detect any differences in size or shape between the two groups (Fig. 2b). Gene expression pattern analysis for the pluripotency marker OCT4 showed no significant differences in differentiation efficiency between conditions. OCT4 is a core transcription factor that maintains pluripotency in iPSCs, and its downregulation during early differentiation serves as a reliable indicator of exit from the pluripotent state. In line with our findings, studies benchmarking pluripotency markers similarly show that OCT4 expression typically declines from day 5 upon germ layer induction, reflecting the onset of lineage commitment30.
The mesoderm marker TWIST indicated normal development of the mesoderm. The expression pattern of mature endothelial and mural cell markers, CD31 and PDGFrβ, respectively, indicated the formation of a similar end-product (Fig. 2c). Additional markers for the different stages (Nanog, SNAIL, SLUG, VE-cadherin, and ACTA2) were also tested, showing similar results (Supplementary Fig. S3b). Surface area quantification based on brightfield imaging revealed no significant differences in size at all timepoints (Fig. 2d). FACS analysis was performed on both groups on day 18. There were no significant differences detected in the composition of the vascular organoids (Fig. 2e). Whole-mount staining confirmed similar structures in both Matrigel-based and Vitronectin-based vascular organoids (Fig. 2f). iPSC clones 1 and 2 demonstrate similar results compared to clone A (Supplementary Fig. S2).
Vascular organoids derived from Vitronectin-based hiPSCs show a similar differentiation pattern compared to Matrigel based hiPSCs. Data shown are from Clone A. (a) Schematic timeline of the differentiation process for both vascular organoid groups. (b) Brightfield images of the vascular organoids during the differentiation process (day 3–18). Scale bar depicts 200 μm. (c) Gene expression analysis of vascular organoids at different timepoints. Results represented as mean ± SEM, n = 4 for Matrigel-based vascular organoids and n = 6 for Vitronectin-based vascular organoids. One-way ANOVA with Tukey post hoc test. No significant differences between the vascular organoid groups were detected. (d) Surface area over time (day 3–18). n = 24 vascular organoids for both groups, 4 technical replicates. Results represented as mean ± SEM. Unpaired T-test between timepoints, no significant differences between the vascular organoid groups were detected. (e) Vascular organoid composition based on FACS results. Cell content is represented as the percentage of total cells measured. Results represented as mean ± SEM, n = 4 for Matrigel-based vascular organoids and n = 6 for Vitronectin-based vascular organoids. Unpaired T-test, no significant differences between vascular organoid groups detected. (f) Whole-mount staining of vascular organoids. Vascular organoids are stained for endothelial cell marker CD31 (Red) and mural cell marker CD140b (Green). Scale bar depicts 200 μm for top panels (whole vascular organoid) and 50 μm for bottom panels (zoomed-in version).
Vascular organoids embedded in a fibrin gel show similar differentiation characteristics to vascular organoids embedded in Matrigel
After switching the hiPSCs from Matrigel to Vitronectin, all vascular organoids for further experiments were cultured on Vitronectin and differentiated as previously described. After 13 days, one group was embedded in a Matrigel-collagen gel, and the other group was embedded in a fibrin gel, as shown in the schematic timeline in Fig. 3a. The fibrin gel was composed of 7.5 mg/mL of fibrinogen together with 0.1 U/mL thrombin. Brightfield images show a similar growth pattern in both conditions from day 13 (Fig. 3b). After 15 days of differentiation, both conditions facilitated vascular organoid sprouting. Gene expression analysis comparing both groups during differentiation showed no significant differences in the expression of endothelial and mural cell markers between fibrin-based and Matrigel-based vascular organoids, except for VE-Cadherin, with significant difference between Matrigel embedded clone 1 and 2 on day 18 (p = 0.0196) (Fig. 3c). As expected, both groups displayed a decrease in mesoderm expression after embedding (Supplementary Fig. S4b). Surface area quantification of the brightfield images confirmed these findings, with no significant differences in the growth pattern up to day 18 (Fig. 3d). FACS analysis of 18-day old vascular organoids showed no significant differences in vascular organoid composition, indicating that both ECM-like gels facilitate vascular organoid development (Fig. 3e). Whole-mount staining of sprouting fibrinogen vascular organoids on day 18 and 21 indicates the development of vascular structures composed of both endothelial and mural cells in the fibrin group, similar to the Matrigel control group (Fig. 3f versus Fig. 2f). Clone A produced similar results compared to Clone 1 and 2, there was no significant differences in growth patterns, gene expression or vascular composition (Supplementary Fig. S5). These results demonstrate that hiPSC differentiation into vascular organoids can be achieved using Vitronectin and fibrin as an effective alternative to Matrigel.
Vascular organoids embedded in fibrin gel show a similar differentiation pattern compared to vascular organoids embedded in Matrigel/collagen. Data shown are from Clones 1 and 2. (a) Schematic timeline of the differentiation process for both vascular organoid groups. (b) Brightfield images of the vascular organoids during the differentiation process (day 13–18). Scale bar depicts 200 μm. (c) Gene expression analysis of vascular organoids at different timepoints. Results represented as mean ± SEM, n = 5 for both groups. One-way ANOVA with Tukey post hoc test. No significant differences between the vascular organoid groups were detected except for VE-Cadherin (p ≤ 0.05). (d) Surface area over time (day 13–18). n = 20 vascular organoids for both groups, 4 technical replicates. Results represented as mean ± SEM. Unpaired T-test between timepoints, no significant differences between the vascular organoid groups were detected. (e) Vascular organoid composition based on FACS results. Cell content is represented as the percentage of total cells measured. Results represented as mean ± SEM, n = 5 for both groups. Unpaired T-test, no significant differences between the vascular organoid groups were detected. (f) Whole-mount staining of vascular organoids. Vascular organoids are stained for endothelial cell marker CD31 (Red) and mural cell marker CD140b (Green). Scale bar depicts 50 μm.
Discussion
Vascular organoids, first described in 2019, demonstrate principles of developmental biology and self-organization, resulting in the first multi-cell lineage human blood vessel networks generated from a single hiPSC29. The original protocol was later validated using multiple cell lines, including those used in this study. The authors noted that slight alterations in culture media and protocols, due to differences in suppliers or batches, can result in ineffective differentiation and altered outcomes. Matrigel, despite being versatile and widely used, is a complex hydrogel composed of over 1800 proteins. It exhibits significant batch-to-batch variation in composition and mechanical properties, which may impact cell and organoid growth31,32,33,34,35. Due to Matrigel’s complexity, animal origins, and high variability, obtaining approval for its use in good manufacturing practice (GMP) laboratories or clinical applications is nearly impossible. As a result, there is a critical need for Matrigel-independent culture systems to improve standardization in organoid-based human models in biomedical research, particularly in regenerative therapies.
HiPSCs cultured on Vitronectin-coated substrate yield similar pluripotent characteristics
Multiple animal-free matrices, such as laminin, fibronectin, and Vitronectin, are reported to support human iPSC and ESC growth. For large-scale applications, Vitronectin is the most cost-effective alternative, with various types available commercially36. Vitronectin, a glycoprotein synthesized in the liver and found in blood and the ECM, is used in this study as a recombinant human protein recognized by cells expressing Vitronectin receptors αvβ1, αvβ3, αvβ5, or αIIbβ3. Although Vitronectin does not form structural networks like Matrigel or fibrin, it promotes cell adhesion and structural integrity by binding to other ECM proteins and integrins. Studies have shown that, when combined with a ROCK inhibitor, Vitronectin supports the initial attachment and survival of human ES and iPSCs as effectively as Matrigel11,37,38. Despite these findings, tumor-derived Matrigel remains widely used in hiPSC differentiation and maintenance protocols. Consistent with previous reports, we demonstrated that culturing hiPSCs on Vitronectin maintained pluripotency without compromising the quality compared to cells cultured on traditional Matrigel.
Vascular organoids derived from Vitronectin-based hiPSCs show a similar differentiation pattern compared to Matrigel-based hiPSCs
ECM-based substrates used for hiPSC expansion may also affect the subsequent differentiation process and potentially alter the function and/or phenotype of the hiPSC-derived tissue cells39,40,41,42. To assess the effect of Vitronectin as an alternative substrate for hiPSC expansion on vascular organoid differentiation, we tested the differentiation protocol described by Wimmer et al.28,29 using hiPSCs grown on Vitronectin versus Matrigel. During the initial differentiation phase, including aggregate formation and size measurements, no significant differences were observed. Further analysis of the differentiation stages via qPCR revealed similar gene expression patterns, with consistent decreases in hiPSC pluripotency markers and mesoderm formation, and an increase in vascular cell development over time in both groups. Assessment of the final product by FACS analysis and whole-mount staining showed comparable cell-type distributions and the formation of endothelial-based vascular structures supported by mural cells. These results indicate that the use of Vitronectin during hiPSC expansion does not affect the differentiation process of subsequent hiPSC-derived vascular organoids.
Vascular organoids embedded in a fibrin gel show similar differentiation characteristics to vascular organoids embedded in Matrigel
In recent years, numerous studies have focused on replacing Matrigel to move towards animal-free culture systems using decellularized ECM, gel-forming recombinant proteins, or synthetic hydrogels for organoid culture. For example, collagen has been utilized for intestine, kidney, colon, and stomach organoids43,44,45, alginate for PSC-derived intestinal organoids46,47, and fibrin-laminin for ASC-derived small intestinal, pancreatic, and liver organoids23. As chemical alternatives, PEG and PGLA have been employed as ECM substitutes for cardiac, intestinal, and lung organoids48,49,50,51,52. These studies demonstrate that the hydrogel characteristics required for organoid development vary based on the organoid type, composition, and intended application, with mechanical properties being a critical consideration. To our knowledge, no animal-free alternatives have been described for organoids containing both endothelial cells and vascular structures. However, successful co-cultures of endothelial cells with supporting cells—such as pericytes or vascular smooth muscle cells (VSMCs)—and sprouting assays have been conducted using animal-free gels. In particular, both collagen and fibrin gels, with or without growth factor supplementation, have been reported to support sprout formation in in vitro angiogenesis models53,54,55,56,57. Fibrin, a key component of the blood-clotting cascade and unsuitable for direct implantation, offers advantageous gel characteristics, a (recombinant) human origin, and lower complexity compared to Matrigel, making it useful for disease modelling and therapeutic screening. It is also suitable for ex vivo differentiation and the culture of hiPSC-derived vascular organoids in biomedical and regenerative applications. Fibrin’s dynamic, porous fibrous mesh facilitates cell migration and infiltration, with fibers that are shorter and thinner than those in collagen gels. Cell interactions occur via integrins such as α5β1, αvβ1, αvβ3, and αvβ5. The stiffness of fibrin can be tailored by adjusting the concentration24. In this study, hiPSCs grown on Vitronectin were assessed for their differentiation efficacy into vascular organoids using two different substrates: Matrigel and a human recombinant fibrin hydrogel (7.5 mg/mL fibrinogen and 0.1 U/mL thrombin). The findings showed no significant differences in differentiation efficacy concerning gene expression patterns, vascular organoid size, or final composition, thus demonstrating that fibrin hydrogel is a viable alternative to Matrigel for the differentiation of Vitronectin-cultured hiPSCs into vascular organoids. In addition, commercial alternative animal-free hydrogels, including GrowDex® hydrogel, VitroGel® and Manchester BIOGEL, were also previously tested in our pipeline, but did not support organoid survival or vascular sprouting, highlighting fibrin as a robust and reproducible matrix for human vascular organoid culture.
We successfully eliminated Matrigel from our hiPSC-derived vascular organoid protocol by developing an animal-free culture system. The morphology and growth efficacy of hiPSCs cultured on Vitronectin were comparable to those grown on Matrigel. Furthermore, vascular organoid differentiation was unaffected by the substrate change. Subsequent hiPSC differentiation using fibrin-based hydrogel enhanced vascular network formation and facilitated endothelial cell sprouting. No significant differences were observed in the resulting vascular organoids compared to the use of Matrigel. These findings demonstrate that Matrigel is not essential for generating vascular organoids. The combination of hiPSC expansion on human recombinant Vitronectin and differentiation on human recombinant fibrin provides a viable alternative to animal-derived components. Based on these findings, we conclude that our Vitronectin and fibrin-based protocol enhances the human relevance and standardization of hiPSC-derived vascular organoids in disease modelling and drug testing.
Vitronectin has been used to derive and expand hiPSCs under chemically defined, xeno-free conditions11 and in neuronal differentiation protocols58. Fibrin has been applied as a 3D scaffold for vascular organoids in an organ-on-a-chip model59 though this remains uncommon. Most studies primarily use fibrin hydrogels for other stem cell applications, such as iPSC-derived retinal pigment epithelium cultures60. To our knowledge, this is the first study to combine recombinant human Vitronectin and recombinant human fibrin into a single xeno-free culture system for vascular organoids. This approach strategically integrates Vitronectin’s support for robust hiPSC expansion with fibrin’s physiologically relevant 3D microenvironment, enabling vascular morphogenesis and long-term organoid stability under fully human-specific conditions. The exclusive reliance on recombinant human proteins eliminates animal-derived variability, thereby improving reproducibility and translational potential. This integrated strategy introduces a novel platform for vascular organoid culture with broad applicability in regenerative medicine, disease modeling, and preclinical testing.
Methods
HiPSC and vascular organoid culture
Derivation and culture of human iPSCs on matrigel and vitronectin
The hiPSC lines used in this study, UKKi032-C (NP0141-31B, referred to as Clone 1) (female) and UKKi036-C (NP0143-18, referred to as Clone 2) (male) are stored in the European Bank for iPSCs (EBiSC; https://ebisc.org/) and registered in the online registry for human PSC lines (hPSCreg; https://hpscreg.eu/). The hiPSC cell lines were generated from peripheral blood mononuclear cells by Sendai viral reprogramming. Donors gave informed consent and the experiments were performed in compliance with the code for proper use of human tissue used in the Netherlands and was approved by the ethics committee of the University Medical Centre Utrecht, the Netherlands. Cell line SCVl273 (referred to as Clone A) was used for Figs. 1 and 2, and Supplementary Fig. S3 and S5 and was donated by the Joseph Wu Lab (Stanford Medicine, Department of Medicine and Radiology, Stanford CVI biobank).
Cells were thawed and cultured on Corning® Matrigel® (1.2 mg/mL in E8 medium, Corning) coated 6-well plates for the Matrigel controls. For the Vitronectin group, cells were thawed and cultured on Vitronectin XF™ (10 µl/mL in PBS, StemCell Technologies) coated 6-well plates. Both groups were supplemented with E8 + medium (Gibco) with 1:1000 ROCK inhibitor (Calbiochem) for the first 24 h. Medium was changed every other day.
HiPSCs in both groups were nonenzymatically passaged every 4 to 5 days at 90% confluence using 0.5 × 10−3 m EDTA (Invitrogen) with a splitting ratio of 1:10. Medium was changed every other day. To improve cell survival, split ratio reliability, and to reduce selective pressure, 1:2000 ROCK inhibitor (Calbiochem) was used in the first 24 h. Cells were passaged at least twice before proceeding to vascular organoid differentiation.
Vascular organoid differentiation – suspension culture
Schematic overview of the experimental procedures is represented in Fig. 2. Both groups of hiPSCs were harvested using EDTA and subsequently resuspended in differentiation medium [Knock-out Dulbecco’s modified Eagle’s culture medium (KO-DMEM): 20% KnockOut Serum (KOSR), 1% Penicillin Streptomycin (PS), 1% Glutamax, and 1% non-essential amino acids (NEAA), all Gibco], including 1:1000 ROCK-inhibitor Y-27632 (Calbiochem). Cells were plated into an ultra-low attachment 6-well plate (Corning), in a final concentration of approximately 2 × 105 cells per well. Cells were incubated on a shaker plate at 37 °C for 24 h to form aggregates and subsequently transferred to hypoxic conditions (5% O2). Cell aggregates were treated with 13 × 10−6 m GSK-3 inhibitor CHIR99021 (Tocris) on day 3 to start mesoderm induction. On days 5, 7 and 9, the aggregates were treated with vascular endothelial growth factor A (VEGF-A) (30 ng/mL; Peprotech), fibroblast growth factor 2 (FGF-2) (30 ng/mL; Miltenyi Biotec), and bone morphogenetic protein 4 (BMP4) (30 ng/mL; StemCell Technologies) to promote vascular Lineage formation. On day 11, differentiation medium was supplemented with VEGF-A (30 ng/mL), FGF-2 (30 ng/mL), and transforming growth factor β (TGFβ)-inhibitor SB43152 (10 × 10−6 m; StemCell Technologies) to increase the yield of endothelial cells and suppress excessive differentiation into mesenchymal/mural like cells.
Vascular organoid differentiation – 3D culture
In this study, 3 different hydrogel regimes were evaluated, displayed in Table 1.
The aggregates were collected on day 13, embedded in a 1:1 Matrigel: collagen mixture (Matrigel-Matrigel group and Vitronectin-Matrigel group) (Corning Matrigel, phenol red free and Atelocollagen Bovine Dermis 3 mg/mL, Bio-connect) or in a 7.5 mg/mL fibrinogen (Gibco) + 0.1 U/mL thrombin (Sigma) gel (Vitronectin-Fibrin group) and supplemented with differentiation medium containing 15% KOSR, VEGF-A (100 ng/mL) and FGF-2 (100 ng/mL). Medium was changed every other day. Vascular organoids were collected on day 18 and either disaggregated for further experiments or cultured in a 96-well round bottom plate without ECM support to stimulate self-assembly into spheroid-shaped vascular organoids.
Cell sorting and analysis
Vascular organoid dissociation
Vascular organoids from both groups were mechanically disrupted after 18 days and disaggregated using 3 U/mL dispase (Gibco), 2 U/mL liberase (Roche) and 100 U DNase (StemCell Technologies) in warm DMEM + Penicillin-Streptomycin (PS) for 20 min at 37 °C while rotating. Every 5 min, the cells were resuspended. To remove excess gel and remaining aggregates from the suspension, the solution was filtered using a 70 μm cell strainer and 5 mL DMEM + PS and subsequently spun down for 5 min at 400 g.
Flow cytometry
Single cell vascular organoids were plated into a 96-well flat bottom plate, approximately 2.5 × 104 cells per well. Cells were washed with PBS supplemented with 2% FBS and 2 mM NaN3. Cells were stained with anti-CD31 and anti-CD140b antibodies (Supplementary Table S1), together with Sytox blue (Invitrogen) to exclude dead cells. CytoFLEX flow cytometer (Beckman Coulter) was used for cell analysis, and data analysis was performed using FlowJo software (Version 10.9.0) and analyzed using GraphPad Prism (Version 10.1.2).
Quantitative polymerase chain reaction (qPCR) analysis
Total RNA was isolated from hiPSCs and vascular organoids on days 0, 5, 11, 15 and 18 of differentiation using Trizol (Invitrogen) according to the manufacturer’s guidelines. The purity and concentration of RNA were quantified by measuring the absorbance at 260/280 nm using spectrophotometry (DS-11; DeNovix). cDNA synthesis was performed according to the Bioline cDNA synthesis kit. Gene expression patterns were determined using FastStart SYBR-green (Roche) following the quantitative polymerase chain reaction (qPCR) program: 8,5′ 95 °C, 38 cycles (15′′ 95 °C; 45′′ 60 °C) 1′ 95 °C, 1′ 65 °C, 62 cycles (10′′ 65 °C + 0.5 °C) in the SYBR-Green-Cycler IQ5 detection protocol (BioRad CFX384), performed in 384-well plates (Merck). The primer sequences used are listed in Supplementary Table S2. All results were normalized to the housekeeping genes β-actin and RPLP0, resulting in relative mRNA expression. Results are shown as the fold change of the Δct values.
Immunohistochemistry and imaging
Immunofluorescent staining of 2D HiPSC cultures
Cultured hiPSCs were fixed using paraformaldehyde (4%) for 15 min and incubated with blocking/permeabilization buffer (5% BSA/0.3% Triton-X-100 in PBS) for 30 min. After blocking with 1:5 diluted blocking buffer (DPBS), the primary antibodies (Supplementary Table S3) were added and incubated overnight at 4 °C. After washing with PBS, Alexa-conjugated secondary antibodies (Life Technologies) (Supplementary Table S3) diluted in 1:5 diluted blocking/permeabilization buffer were incubated for one hour in the dark at room temperature (RT). Cell nuclei were visualized using Hoechst or DAPI (ThermoFisher) before imaging with a Leica DMi8 confocal microscope or Nikon Ti2 Widefield microscope (Trilineage).
Whole-mount staining of 3D vascular organoid cultures
Vascular organoids were extracted from wells or a 96-well plate and fixed for 2 h with 4% PFA at RT. Vascular organoids were blocked and permeabilized using 3% FBS, 1% BSA, 0.5% Triton x-100, and 0.5% Tween in PBS for 2 h at RT. The 3D vascular organoids were stained with anti-CD31 and anti-PDGFrβ antibodies (Supplementary Table S3) for 2 h at RT. The vascular organoids were washed three times with PBS-/Tween (PBST), and secondary antibody incubation (Supplementary Table S3) was performed for 2 h at RT. Vascular organoids were washed again three times with PBST. DAPI was used as a counterstain, and 3D cell cultures were mounted with Mowiol4-88. Samples were stored at 4 °C prior to imaging.
Imaging and analysis
Brightfield images for surface area analysis were taken using the Olympus CKX41. 3D Imaging was performed using the Leica Confocal SP8 × (10×, 20× magnifications). Images were analyzed using ImageJ software (Version 1.47). 3D images were composed in LASX (Version 3.5.7.23225).
Statistical analysis
Statistical analyses were performed using GraphPad Prism (Version 10.1.2). Values are shown as individual data points with mean ± SEM. Prior to statistical testing, outliers were removed if detected using a Grubbs’ test (alpha = 0.05). The paired, two-sided t-test and ordinary one-way ANOVA test with Tukey post hoc test were used when appropriate. Experiments were performed at least in triplicate. The detailed sample size for each result is listed in the figure legend. A p-value of p ≤ 0.05 was accepted as statistically significant.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information files. Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request.
References
-
Kozlowski, M. T., Crook, C. J. & Ku, H. T. Towards organoid culture without Matrigel. Commun. Biol. https://doi.org/10.1038/s42003-021-02910-8 (2021).
-
Hughes, C. S., Postovit, L. M. & Lajoie, G. A. Matrigel: a complex protein mixture required for optimal growth of cell culture. Proteomics 10, 1886–1890 (2010).
-
Passaniti, A., Kleinman, H. K. & Martin, G. R. Matrigel: history/background, uses, and future applications. J. Cell Commun. Signal. 16, 621–626. https://doi.org/10.1007/s12079-021-00643-1 (2022).
-
Heo, J. H., Kang, D., Seo, S. J. & Jin, Y. Engineering the extracellular matrix for organoid culture. Int. J. Stem Cells. 15, 60–69 (2022).
-
Kleinman, H. K., Philp, D. & Hoffman, M. P. Role of the extracellular matrix in morphogenesis. Curr. Opin. Biotechnol. 14, 526–532. https://doi.org/10.1016/j.copbio.2003.08.002 (2003).
-
Kleinman, H. K. & Martin, G. R. Matrigel: basement membrane matrix with biological activity. Semin. Cancer Biol. 15, 378–386. https://doi.org/10.1016/j.semcancer.2005.05.004 (2005).
-
Rodin, S. et al. Long-term self-renewal of human pluripotent stem cells on human Recombinant laminin-511. Nat. Biotechnol. 28, 611–615 (2010).
-
Pouliot, N. & Kusuma, N. Laminin-511: A multi-functional adhesion protein regulating cell migration, tumor invasion and metastasis. Cell Adhesion Migr. 7, 142–149. https://doi.org/10.4161/cam.22125 (2013).
-
Miyazaki, T. et al. Recombinant human laminin isoforms can support the undifferentiated growth of human embryonic stem cells. Biochem. Biophys. Res. Commun. 375, 27–32 (2008).
-
Lam, M. T. & Longaker, M. T. Comparison of several attachment methods for human iPS, embryonic and adipose-derived stem cells for tissue engineering. J Tissue Eng. Regen Med https://doi.org/10.1002/term.1499 (2012).
-
Chen, G. et al. Chemically defined conditions for human iPSC derivation and culture. Nat. Methods. 8, 424–429 (2011).
-
Shen, J. et al. Vitronectin-activated αvβ3 and αvβ5 integrin signalling specifies haematopoietic fate in human pluripotent stem cells. Cell Prolif. 54, e13012 (2021).
-
Fontes, A., MacArthur, C. C., Lieu, P. T. & Vemuri, M. C. Generation of human-induced pluripotent stem cells (hiPSCs) using episomal vectors on defined essential 8™ medium conditions. Methods Mol. Biol. 997, 57–72 (2013).
-
Aisenbrey, E. A. & Murphy, W. L. Synthetic alternatives to Matrigel. Nat. Rev. Mater. 5, 539–551. https://doi.org/10.1038/s41578-020-0199-8 (2020).
-
Li, C. et al. Enhancing organoid culture: Harnessing the potential of decellularized extracellular matrix hydrogels for mimicking microenvironments. J. Biomed. Sci. 31, 96 (2024).
-
Marchini, A. & Gelain, F. Synthetic scaffolds for 3D cell cultures and organoids: applications in regenerative medicine. Crit. Rev. Biotechnol. 42, 468–486 (2022).
-
Morin, K. T. & Tranquillo, R. T. Guided sprouting from endothelial spheroids in fibrin gels aligned by magnetic fields and cell-induced gel compaction. Biomaterials 32, 6111–6118 (2011).
-
Nguyen, E. H. et al. Versatile synthetic alternatives to matrigel for vascular toxicity screening and stem cell expansion. Nat. Biomed. Eng. https://doi.org/10.6084/m9.figshare.c (2017).
-
Lust, S. T. et al. Selectively Cross-Linked Tetra-PEG hydrogels provide control over mechanical strength with minimal impact on diffusivity. ACS Biomater. Sci. Eng. 7, 4293–4304 (2021).
-
Morin, K. T. & Tranquillo, R. T. In vitro models of angiogenesis and vasculogenesis in fibrin gel. Exp. Cell Res. 319, 2409–2417. https://doi.org/10.1016/j.yexcr.2013.06.006 (2013).
-
Nakatsu, M. N., Davis, J. & Hughes, C. C. W. Optimized fibrin gel bead assay for the study of angiogenesis. J. Visualized Experiments. https://doi.org/10.3791/186 (2007).
-
Nakatsu, M. N. et al. Angiogenic sprouting and capillary lumen formation modeled by human umbilical vein endothelial cells (HUVEC) in fibrin gels: the role of fibroblasts and Angiopoietin-1. Microvasc Res. 66, 102–112 (2003).
-
Brinkmann, J. et al. Assessment of Fibrin-Based Hydrogels Containing a Fibrin-Binding Peptide to Tune Mechanical Properties and Cell Responses. Macromol Mater. Eng 308, 2200678 (2023).
-
Janmey, P. A., Winer, J. P. & Weisel, J. W. Fibrin gels and their clinical and bioengineering applications. J. Royal Soc. Interface 6, 1–10 (2009).
-
Broguiere, N. et al. Growth of epithelial organoids in a defined hydrogel. Adv. Mater. 30, 1801621 (2018).
-
Murphy, K. C., Whitehead, J., Zhou, D., Ho, S. S. & Leach, J. K. Engineering fibrin hydrogels to promote the wound healing potential of mesenchymal stem cell spheroids. Acta Biomater. 64, 176–186 (2017).
-
Chung, E. et al. Fibrin-based 3D matrices induce angiogenic behavior of adipose-derived stem cells. Acta Biomater. 17, 78–88 (2015).
-
Meijer, E. M. et al. 3D Human iPSC Blood Vessel Organoids as a Source of Flow-Adaptive Vascular Cells for Creating a Human-Relevant 3D-Scaffold Based Macrovessel Model. Adv Biol 7, 2200137 (2023).
-
Wimmer, R. A. et al. Human blood vessel organoids as a model of diabetic vasculopathy. Nature 565, 505–510 (2019).
-
Cevallos, R. R., Hossain, M. E., Zhang, R. & Hu, K. Evaluating reprogramming efficiency and pluripotency of the established human iPSCS by pluripotency markers. Methods Mol. Biol. 2239, 235–249 (2021).
-
Vukicevic, S. et al. Identification of multiple active growth factors in basement membrane matrigel suggests caution in interpretation of cellular activity related to extracellular matrix components. Exp. Cell. Res. 202, 1–8 (1992).
-
Huch, M., Knoblich, J. A., Lutolf, M. P. & Martinez-Arias, A. The hope and the hype of organoid research. Dev. (Cambridge). 144, 938–941 (2017).
-
Nemir, S. & West, J. L. Synthetic materials in the study of cell response to substrate rigidity. Ann. Biomed. Eng. 38, 2–20 (2010).
-
Chaudhuri, O. Viscoelastic hydrogels for 3D cell culture. Biomater. Sci. 5, 1480–1490. https://doi.org/10.1039/c7bm00261k (2017).
-
Dahl-Jensen, S. & Grapin-Botton, A. The physics of organoids: A biophysical approach to Understanding organogenesis. Dev. (Cambridge). 144, 946–951 (2017).
-
Braam, S. R. et al. Recombinant vitronectin is a functionally defined substrate that supports human embryonic stem cell Self-Renewal via αVβ5 integrin. Stem Cells. 26, 2257–2265 (2008).
-
Rowland, T. J. et al. Roles of integrins in human induced pluripotent stem cell growth on matrigel and vitronectin. Stem Cells Dev. 19, 1231–1240 (2010).
-
Rivera, T., Zhao, Y., Ni, Y. & Wang, J. Human-Induced Pluripotent Stem Cell Culture Methods Under cGMP Conditions. Curr Protoc. Stem Cell. Biol 54, e117 (2020).
-
LaFosse, P. et al. Effect of substrate coating material on spontaneous activity of human-induced pluripotent stem cell-derived neuronal stem cells. Front Cell. Neurosci https://doi.org/10.3389/conf.fncel.2018.38.00115 (2018).
-
Lee, C. H. et al. Tuning pro-survival effects of human induced pluripotent stem cell-derived exosomes using elastin-like polypeptides. Biomaterials 291, 121864 (2022).
-
Sağraç, D. et al. Surface coating materials regulates the attachment and differentiation of mouse embryonic stem cell derived embryoid bodies into mesoderm at culture conditions. Cytotechnology 74, 293–307 (2022).
-
Prajapati, C., Ojala, M., Lappi, H., Aalto-Setälä, K. & Pekkanen-Mattila, M. Electrophysiological evaluation of human induced pluripotent stem cell-derived cardiomyocytes obtained by different methods. Stem Cell. Res 51, 102176 (2021).
-
Jabaji, Z. et al. Use of collagen gel as an alternative extracellular matrix for the in vitro and in vivo growth of murine small intestinal epithelium. Tissue Eng. Part. C Methods. 19, 961–969 (2013).
-
Sachs, N., Tsukamoto, Y., Kujala, P., Peters, P. J. & Clevers, H. Intestinal epithelial organoids fuse to form self-organizing tubes in floating collagen gels. Dev. (Cambridge). 144, 1107–1112 (2017).
-
Jee, J. H. et al. Development of Collagen-Based 3D Matrix for Gastrointestinal Tract-Derived Organoid Culture. Stem Cells Int. 2019, 8472712 (2019).
-
Capeling, M. M. et al. Nonadhesive alginate hydrogels support growth of pluripotent stem Cell-Derived intestinal organoids. Stem Cell. Rep. 12, 381–394 (2019).
-
Patel, S. N. et al. Organoid Microphysiological System Preserves Pancreatic Islet Function within 3D Matrix. Sci. Adv. https://doi.org/10.1126/sciadv.aba5515 (2021).
-
Cruz-Acuña, R. et al. PEG-4MAL hydrogels for human organoid generation, culture, and in vivo delivery. Nat. Protoc. 13, 2102–2119 (2018).
-
Hoang, P. et al. Engineering spatial-organized cardiac organoids for developmental toxicity testing. Stem Cell. Rep. 16, 1228–1244 (2021).
-
Cruz-Acuña, R. et al. Synthetic hydrogels for human intestinal organoid generation and colonic wound repair. Nat. Cell. Biol. 19, 1326–1335 (2017).
-
Dye, B. R. et al. Human lung organoids develop into adult airway-like structures directed by physico-chemical biomaterial properties. Biomaterials 234, 119757 (2020).
-
Davoudi, Z. et al. Intestinal organoids containing poly(lactic-co-glycolic acid) nanoparticles for the treatment of inflammatory bowel diseases. J. Biomed. Mater. Res. A. 106, 876–886 (2018).
-
Dietrich, F. & Lelkes, P. I. Fine-tuning of a three-dimensional microcarrier-based angiogenesis assay for the analysis of endothelial-mesenchymal cell co-cultures in fibrin and collagen gels. Angiogenesis 9, 111–125 (2006).
-
Rioja, A. Y., Annamalai, T., Paris, R., Putnam, S., Stegemann, J. P. & A. J. & Endothelial sprouting and network formation in collagen- and fibrin-based modular microbeads. Acta Biomater. 29, 33–41 (2016).
-
Feng, X., Tonnesen, M. G., Mousa, S. A. & Clark, R. A. F. Fibrin and collagen differentially but synergistically regulate sprout angiogenesis of human dermal microvascular endothelial cells in 3-dimensional matrix. Int. J. Cell. Biol. https://doi.org/10.1155/2013/231279 (2013).
-
Lawley, T. & Kubota, Y. Induction of morphologic differentiation of endothelial cells in culture. J. Investig. Dermatol. https://doi.org/10.1038/jid.1989.10 (1989).
-
Kanzawa, S., Endo, H. & Shioya, N. Improved in vitro angiogenesis model by collagen density reduction and the use of type III collagen. Ann. Plast. Surg. 30, 244–251 (1993).
-
Robinson, M., Chapani, P., Styan, T., Vaidyanathan, R. & Willerth, S. M. Functionalizing Ascl1 with novel intracellular protein delivery technology for promoting neuronal differentiation of human induced pluripotent stem cells. Stem Cell. Rev. Rep. 12, 476–483 (2016).
-
Quintard, C. et al. A microfluidic platform integrating functional vascularized organoids-on-chip. Nat. Commun. 15, 1452 (2024).
-
Gandhi, J. K. et al. Fibrin hydrogels as a xenofree and rapidly degradable support for transplantation of retinal pigment epithelium monolayers. Acta Biomater. 67, 134–146 (2018).
Funding
This work was funded by the RegMed XB cardiovascular moonshot consortium and the NWO vidi grant (no. 91714302 to Caroline Cheng). The authors acknowledge the Gravitation Program “Materials Driven Regeneration”, funded by the Netherlands Organization for Scientific Research (024.003.013). The authors also acknowledge support from the Dutch CardioVascular Alliance (an initiative with support from the Dutch Heart Foundation) Grant 2020B008 Reconnext (to Rachel Giles, Christian G.M. van Dijk, Marianne C. Verhaar and Caroline Cheng).
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Giles, R., Meijer, E.M., Maas, R.G. et al. Animal-free alternatives for Matrigel in human iPSC-derived blood vessel organoid culture. Sci Rep 15, 36042 (2025). https://doi.org/10.1038/s41598-025-20091-w
-
Received:
-
Accepted:
-
Published:
-
DOI: https://doi.org/10.1038/s41598-025-20091-w