- Review
- Open access
- Published:
- Yongchen Tai1,
- Yunshen Li1,
- Kayla M. Mornay2,
- Madison K. Woodard2,
- Wenting Wang1,
- Xianrui Yang2 &
- …
- Jing Pan1
npj Biosensing volume 3, Article number: 14 (2026) Cite this article
Subjects
Abstract
Oral and dental health is an important indicator and determinant of an individual’s overall well-being. Untreated oral diseases can lead to severe systemic complications. Monitoring the oral environment and identifying biochemical and physiological patterns associated with disease states, such as periodontitis, gingivitis, caries, and oral cancers, is essential for early diagnosis and effective intervention. This review evaluates the current clinical needs in biochemical and physiological monitoring for oral healthcare and state-of-the-art biosensors capable of continuous analyte measurement. We surveyed the relevant biomarkers for common oral and dental diseases in patients compared to healthy controls. The design and performance of recent biosensing devices for these target analytes are reviewed and evaluated. For biochemical sensing, we found intraoral biosensors for high-abundance small molecules, such as ions and metabolites, have advanced significantly in recent years. However, robust sensing technologies for low-abundance analytes, including cytokines and other inflammatory biomarkers, remain limited and require further development in sensing mechanisms, bio-interfaces, and device integration. For physiological sensing, particularly the measurement of forces in tooth movement, recent developments in force sensor technologies have substantially improved measurement accuracy over traditional techniques. Despite these advancements, current platforms still face limitations in achieving long-term, real-time monitoring of mechanical conditions within the oral cavity due to challenges related to biocompatibility and device miniaturization. In conclusion, while notable progress has been made in biosensing for oral applications, continued research in device integration with clinical practices is essential to realize robust and clinically deployable biosensor systems that can advance precision oral healthcare.
Introduction
Oral and dental health is reflective of an individual’s overall health and well-being. When compromised, it can lead to significant systemic complications. In 2025, approximately 3.7 billion people suffered from oral diseases, accounting for nearly half of the global population1. Deteriorating dental and oral health can significantly reduce quality of life and impact confidence2. Diseases of the stomatognathic system include the jaw, teeth, and soft tissues of the oral cavity. These diseases that affect the global population can include periodontal disease, craniofacial anomalies, head and neck cancers, craniofacial benign tumors, caries, and malocclusion2. Monitoring the progression of pathological processes in the mouth is vital for maintaining a stable oral microbiome to prevent the establishment and progression of disease. Despite ongoing efforts to reduce the high prevalence of dental diseases, the oral healthcare system continues to fall short in meeting population needs, limiting access to appropriate dental services, and a population burdened with untreated dental diseases3.
Accurate diagnosis is essential to prevent irreversible oral damage, yet current clinical practice relies on symptomatic patient visits and sporadic2, lab-based diagnostics that are costly, slow, and offer only intermittent snapshots of biomarker levels and disease states. These intermittent measurements generate sparse clinical data that fail to guide timely intervention4 or continuous monitoring. Socioeconomic, systemic, logistical, and geographic barriers5 further delay care, leading to advanced disease stages, patient noncompliance, and suboptimal outcomes. The combined challenges highlight the need for low-cost, user-friendly technologies capable of noninvasive6, continuous biomarker monitoring and personalized oral health management.
Biosensing technology offers a transformative opportunity to overcome current limitations in the clinical management of oral and dental health. Recent advances in portable and wearable biosensors7 have facilitated the integration of sensing elements into the oral environments via intraoral patches8, smart toothbrushes9, mouthguards10,11,12,13, dental implants14,15, and salivary diagnostic devices16,17. While several other notable wearable platforms, such as smart glasses, watches, and contact lenses18 have demonstrated utility in health monitoring, the oral cavity presents a uniquely accessible site rich in biofluids, particularly saliva, which contains biomarkers reflective of both local and systemic physiology. The global demand for biosensing technologies has a projected market value of up to $70 billion USD7,19, which underscores their potential in facilitating the shift from population-centered to patient-centered care. In clinical practice, these devices could enable real-time detection of disease-associated biomarkers, such as inflammatory cytokines, glucose, pH, and bacterial metabolites, allowing early identification of conditions like periodontitis, caries, and systemic metabolic disorders. By capturing patient-specific molecular signatures, biosensing technologies enhance diagnostic accuracy and support timely, targeted interventions, ultimately improving patient outcomes and preventing disease progression.
Despite advances in oral healthcare and sensing technology development, there remains a critical knowledge gap in understanding how biosensing technologies can be effectively applied to address unmet diagnostic and monitoring needs in oral health applications. A systematic review is therefore needed to identify opportunities, challenges, and translational pathways for sensor integration across various oral health applications. This review first examines the clinical needs and technical challenges associated with the application of biosensors in dental, oral, and Craniofacial applications. It then focuses on the use of biosensing technologies for detecting disease-specific biomarkers, with emphasis on their roles in managing periodontal disease, monitoring orthodontic treatment, and supporting early diagnosis of oral cancer and other dental diseases. Additionally, this paper surveys current biosensor design strategies, highlighting key limitations in sensitivity, biocompatibility, and integration within the oral environment. Finally, it explores emerging directions in dental biosensing, outlining opportunities for innovation that can enhance diagnostic precision and therapeutic outcomes.
Methodology
A structured literature search was conducted to identify relevant publications that discussed biosensors in the dental, oral, and craniofacial environments as well as various clinical conditions, such as periodontitis, gingivitis, peri-implantitis, oral cancer, dental fluorosis, caries, edentulism, craniofacial anomalies, malocclusion, and temporo-mandibular disorder (TMD). Electronic databases, including Google Scholar and PubMed, were used to locate relevant systematic reviews, randomized controlled trials, review papers, and case studies.
Keywords such as, “periodontitis”, “periodontal disease”, “gingivitis”, “peri-implantitis”, “oral cancer”, “squamous cell carcinoma”, “fluorosis”, “caries”, “cavities”, “craniofacial anomalies”, “malocclusion”, “temporomandibular disorder”, “TMD”, “dental biosensor”, “biomarkers”, “dental biomarkers”, “biosensors” and “Wearable biosensor” were used individually and in combination for an initial broad search. There was no publication year restriction.
Articles were manually evaluated based on relevance and accessibility of the full text. Priority was given to articles that were published within the past 10 years to ensure accurate data and technologies; however, older studies were also considered when appropriate.
The following inclusion criteria were considered for eligible studies for this review paper: (1) discussion of biosensor technology applied to dental, oral, or craniofacial health; (2) studies available in full text; (3) publications in full English text; and (4) provision of qualitative or quantitative data surrounding biosensors or relevant oral conditions.
Due to this paper being a review, no risk of bias tool was applied; however, data collection and research were conducted in systematically to ensure a thorough and comprehensive scope of knowledge.
Clinical needs and technical challenges
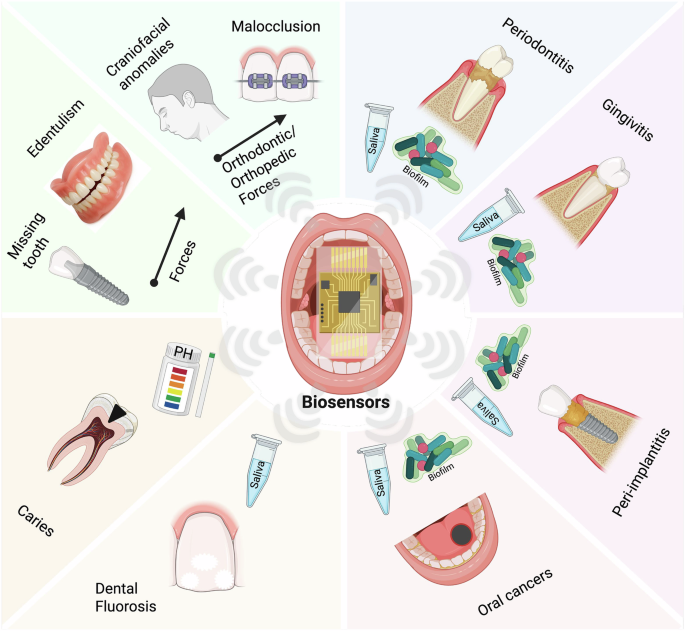
The oral cavity serves as a unique interface between the human body and the external environment, providing direct access to the respiratory and digestive systems, while maintaining close anatomical and functional connections to the circulatory and nervous systems. In addition to its functional and physiological significance, the oral cavity harbors its own microbiome, creating a diverse ecosystem of bacteria, viruses, microeukaryotes, and archaea, all of which play significant roles in oral and overall health20. Disruptions and diseases in this environment are increasingly linked to a heightened risk of systemic conditions, such as cardiovascular disease21. Many diseases in the oral cavity remain undetected until they have progressed to an advanced stage, which often creates severe, even irreversible damage. Microorganisms are the root cause of diseases such as dental caries, periodontal disease, and gingivitis22. Early detection and identification of the infection burden, as well as the continuous tracking of biomolecular changes in the oral environment, such as pH or inflammation, are key for optimal patient prognosis and treatment. For example, studies have reported that simple oral visual screening of the head, neck, and oral cavity to detect the presence of suspicious lesions can reduce mortality in individuals with significant risk factors23. For more thorough inspections, histopathology and diagnostic imaging are commonly adopted in clinical practice but come at a great economic burden to patients23. Additionally, understanding the body’s response to forces or prosthetic interventions, such as the 3D spatiotemporal forces and subsequent mechanotransduction and inflammatory responses, is important for personalized treatment of malocclusions and management of implants and dentures for teeth replacement. These clinical needs highlight the importance of developing advanced biosensing technologies that are efficient, cost-effective, and targeting a diverse range of analytes (Fig. 1). In this section, we review clinically important disease biomarkers and the technical challenges associated with the detection and continuous tracking of these biomarkers.
Biosensors can detect biochemical markers of diseases through biofluids, such as saliva and microbial biofilm. They can also detect changes in pH values and mechanical forces. Clinicians can leverage the sensing data to support the early diagnosis and personalized management of various disease states in the oral cavity, such as excessive inflammation, dental caries, implants, and dentures for missing tooth and edentulism, malocclusions, and craniofacial anomalies.
Infection and inflammation
Microorganisms adhere to the tooth structure and can accumulate into biofilms, which cause dental caries and lead to inflammatory diseases such as gingivitis and periodontitis24. Detecting biochemical markers in the oral environment can help quantify and monitor the infection burden and host response25. Levels of biochemicals, such as cytokines, enzymes, and metabolites, have shown clinical utility in the diagnosis and treatment of various oral diseases26. The biomarkers for dental and oral disease are listed in Table 1.
Periodontitis
Periodontitis affects nearly 42% of the United States, making it one of the most prevalent chronic inflammatory diseases27. The systemic manifestations of periodontitis are associated with a myriad of diseases, such as Alzheimer’s, chronic kidney disease, diabetes, respiratory tract infections, obesity, and even adverse pregnancy outcomes28,29. Clinical signs of periodontitis involve persistent bacterial-induced inflammation, degradation of the periodontal ligament, alveolar bone loss, and eventually tooth loss30. A significant contributor to disease progression is the elevated presence of the Gram-negative red complex bacteria Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia, which are associated with tissue destruction31. Periodontal disease can progress with only mild gum bleeding, making early detection by traditional clinical examination unreliable32. Monitoring the bacterial load and enzymatic markers of disease pathogenesis is needed for effective diagnosis and treatment. Enzymes such as matrix metalloproteinases-8 (MMP-8) and MMP-9, which mediate collagen breakdown, are correlated with disease severity. Studies have shown the levels of MMPs combined with pro-inflammatory cytokines, such as interleukin-1β (IL-1β), can improve the sensitivity in predicting periodontitis by 74%27. Additional biomarkers, including cytokines (e.g., IL-6, IL-8, and TNF-α), enzymes, and proteins involved in bone remodeling (e.g., cathepsin and receptor activator of nuclear factor kappa-B ligand or RANK ligand) are also important biomarkers for clinical assessment and disease management.
Gingivitis
Gingivitis represents an early and potentially reversible stage of periodontitis33,34. It is a disease that involves gingival inflammation due to the accumulation of microbial communities, often resulting from inadequate oral hygiene practices35,36. The presence of infection leads to the recruitment of immune effector cells and the release of pro-inflammatory mediators. Enzymes such as MMPs, alkaline phosphatase (ALP), and acute-phase proteins like C-reactive protein (CRP) are also elevated during gingival inflammation. As gingivitis is a precursor of periodontitis, many of the biomarkers used for periodontitis are also relevant, detectable, and clinically needed in the management of gingivitis.
Peri-implantitis
Dental implants are susceptible to inflammatory conditions that closely resemble diseases affecting natural teeth37. Peri-implantitis has a similar pathogenesis as periodontitis, resulting in the progressive inflammation and degradation of the supporting bone, significantly reducing implant stability38. Due to similar pathological mechanisms, periodontitis and peri-implantitis share many of the same biomarkers. Although clinical diagnosis of peri-implantitis is common, the current diagnostic approach suffers from limited precision and delayed detection, which increases the risk of implant failure39.
Caries
Dental caries is one of the most widespread diseases globally40,41. It develops when Streptococcus mutans metabolizes dietary sucrose, producing lactic acid that lowers pH in the local environment, leading to enamel demineralization and cavity formation25. There is a wide variety of biomarkers identified to indicate the presence of caries activity, such as salivary α-amylase (sAA)25. sAA has been shown to increase in the presence of dental caries and could serve as a diagnostic biomarker for assessing caries risk42. Other biomarkers that can be used for the detection of dental caries include ammonia, urea, pH, and ions like calcium, phosphate, and fluoride. Low pH is a strong indicator of active caries as it results directly from acidogenic bacterial metabolism. Conversely, elevated levels of ammonia or urea are indicative of patients with low-caries activity, as these are protective against the development of caries43.
Oral cancer
Oral cancer, such as squamous cell carcinoma (OSCC), ranks among the top six most prevalent cancers worldwide and has seen an 11% increase in mortality between 1990 and 202144,45. The early symptoms of oral cancer are mild or even absent, so patients may not seek care until the disease is advanced, leading to poor prognosis and survival46. Given its prevalence and aggressive nature, there is a critical need to understand and monitor biomolecular patterns associated with oral cancer that have high specificity and sensitivity. Several biomarkers have shown strong diagnostic potential for OSCC47. IL-8 is among the most studied, with a well-established role in angiogenesis and tumor metastasis47. Elevated levels of IL-8 and IL-1β have demonstrated the ability to distinguish not only between healthy and malignant tissue but also between advanced cancer stages (III vs. IV)48. Similarly, IL-6, which is associated with tumor-promoting signaling pathways, is often elevated in both serum and saliva of OSCC patients49. Additional biomarkers investigated before include MMP-9, which contributes to extracellular matrix degradation and tumor invasion, and lactate dehydrogenase, a metabolic enzyme linked to cell proliferation and membrane disruption in cancerous tissues47. Most available studies rely on single-point or sparse measurements of these disease markers. Because oral cancer progression is a dynamic process, capturing the time-resolved fluctuation of biomarker levels is also clinically needed for early detection, staging, and personalized treatment of oral cancer.
Fluorosis
Fluoride is a nutrient beneficial for healthy growth, but excessive intake over extended periods of time can lead to altered oral microbiome, weakened bone and teeth, and chronic dental and skeletal fluorosis25,50. Recognition of different types of fluorides and compounds, such as CaF2, KF, and SrF2 in water and the oral environment can help monitor dental fluorosis51. Detecting increased presence of ions such as F⁻, Ca²⁺, and PO₄³⁻ could indicate fluorosis is present in the oral environment and help monitor treatment progress.
Malocclusion and craniofacial anomalies
Malocclusion and craniofacial anomalies are managed through orthodontic and orthopedic treatments that rely on controlled force application to guide tooth movement and improve function and esthetics52. Management and monitoring of forces in orthodontics and dentofacial orthopedics is imperative in correcting misalignment of the teeth and jaw. Both the force applied during treatment and patient compliance to treatment protocols are important to achieve optimal outcomes53. For example, bond failure between the orthodontic bracket and the tooth structure may be neglected by patients, which leads to deviation of applied forces from the treatment plan. However, accurate measurement of the 3D loading forces on teeth and longitudinal monitoring of force adaptation during orthodontic movement is technically challenging and not available in clinical practices today. Prolonged excessive forces in orthodontic tooth movement can cause root resorption, tooth mobility, and damage to the alveolar bone, while inadequate force reduces treatment efficacy. For example, in patients with orthodontically induced inflammatory root resorption (OIIRR), they displayed an increase of pro-inflammatory salivary cytokines such as IL-7, and IFN-γ54. Although these cytokines are present during force movements in patients with OIIRR, they can also be induced by other factors, suggesting a need for further research assessing the relationship with these cytokines54. Although multiple pursuits have been made in determining orthodontic forces via animal experiments, in vitro sensor bracket models, and clinical trials, none have obtained accurate measurements52,55.
Besides orthodontic treatments, the development of accurate force sensors is also needed in several other oral and dental health applications. For example, maximum bite force is a useful metric in addition to specific morphological classification of malocclusion to determine the severity and functional outcome of the disease56,57,58,59,60,61. Another clinical need for force measurement is in dental implants. Strong foundational support of the alveolar bone is essential for the anchoring of an implant62. Currently, information regarding bone formation is obtained via radiographic X-ray imaging, which can only be done in a dental clinic and not compatible with continuous monitoring due to radiation exposure. Biosensors integrated with dentures can be used to assess the mechanical fit, stability, and biocompatibility without the use of X-ray imaging63. This will enable wireless and continuous implant information collection, which reduces the number of dental visits, is cost-effective, and could potentially improve the efficacy of dental implants.
Temporomandibular disorder, TMD, is classified as a disorder of the temporomandibular joints in the jaw that can be caused by trauma, injury, malocclusion, or different stress factors. This disorder can cause pain, limitations in opening and closing of the mouth, and dysfunction in daily activities such as chewing, affecting up to 12% of the population. Due to communication with the blood, saliva has shown to display elevated biomarkers associated with TMD, such as salivary cortisol, which is indicated as a stress biomarker for the disorder64. Other studies have found higher levels of salivary and plasma glutamate in patients with TMD, suggesting that the dysregulation of glutamate may be involved in the pathophysiology of TMD65. In some studies, higher levels of IL-1 have also been found in patients with TMD alone, showing a relationship between inflammatory mediators and the disorder66. Saliva has shown to be a non-invasive potential biomarker for TMD; however, continuous monitoring systems have yet to be developed to monitor the disorder. Literature suggests a developing need for biosensors related to the treatment of TMD.
Bruxism
Bruxism is a parafunctional habit in which individuals subconsciously clench their jaw for extended periods of time during their sleep. This habit can result in worn dentition, dysfunction of the temporomandibular joint, pain, and tooth sensitivity. Studies have found that bruxers display higher levels of perceived psychological stress and salivary cortisol levels67. The development of an intraoral continuous monitoring system for the detection of bruxism can help clinicians improve patient outcomes.
Technical challenges
Current medical technology cannot meet the clinical needs due to several technical challenges. First, it is difficult to achieve analytical sensitivity in the physiological range of oral biomarkers under continuous sensor operation. The physiological range of several important biomarkers is very low, while the sample matrix is highly complex. Continuous sensors like continuous glucose monitoring are only available for single analyte and does not work for low-abundance analytes such as cytokines. Second, it is difficult to achieve continuous measurement. Standard immunoassays such as ELISA can precisely quantify the concentration of the analyte. However, the necessity for large assay volumes, manual sampling intervals, and prolonged incubation periods limits the frequency of measurements and introduces delays that prevent continuous measurement. Third, the dynamically changing oral environment makes anti-biofouling difficult. The oral environment can dynamically change depending on oral hygiene, food intake, and other physiological states68,69. This presents unique challenges for the device design of biosensors in the oral cavity. Independent of the signal-transduction mechanism, sensor specificity and selectivity are often dependent on the probe–target interaction and the type of surface passivation employed. Biofouling, such as nonspecific adsorption of random targets and matrix contaminants onto the sensor surface, instability of the immobilized probe, and detachment of the passivation layers, can degrade the sensor specificity and sensitivity and cause signal drift over time. Preventing nonspecific adsorption while withstanding the constantly changing environment is especially essential for developing sensors for dental and oral applications in this complex environment70,71. At the same time, the sensing device needs to be in a minimally invasive, compact format with a sensing probe, data readout, battery, and wireless transmission all integrated. All of these requirements highlight the engineering challenges of interfacing a sensing device in the oral cavity53. Next, we review recent developments in biosensing technologies targeting oral and dental healthcare applications.
Sensors for biochemical monitoring in oral health applications
Bioanalytical sensor design and characterization
Bioanalytical sensors are typically composed of three core components (Fig. 2a): a bio-recognition element that selectively interacts with target analytes72 (Fig. 2b), a signal transduction element that converts the biochemical interaction into a measurable signal (Fig. 2c), and a signal readout element that records or displays the processed output73,74,75,76,77. The bio-recognition elements comprise categories of enzymes11,13,14,17,78, target selective materials16,79,80,81,82, and binding reagents such as aptamers83,84,85,86, antibodies87,88,89,90,91,92, or synthetic binders15,93. Biorecognition signals are commonly transduced and observed either optically by imaging photon changes or electrically by probing electron transfer. Adapting these systems for wearable oral applications requires integration of all components into a compact, biocompatible, and mechanically robust sensor platform capable of maintaining stability and performance under physiological conditions73 (Fig. 2d). Current intraoral biosensor platforms have been integrated into a variety of device formats such as dentures, mouthguards, modified wooden tongue depressors, dental floss, and toothbrushes. Antifouling strategies are employed to allow these platforms to withstand the complex environment, reduce the overtime calibration drift, and prevent nonspecific absorption of complex proteins and contaminations to ensure selectivity and sensitivity. The antifouling strategies include passivation with molecules such as casein90, biotin, molecule monolayers94, or poly (ethylene glycol) (PEG)89, and protecting the sensor through coating a protection layer such as ion-selective membranes16,79,80,81,82. These platforms are calibrated either in actual or artificial saliva and gingival crevicular fluid matrices to correct for matrix effects. These platforms have been employed to detect a broad range of clinically relevant biomarkers. For oral and dental applications, common analytes include glucose, nitrite, lactate, uric acid, thiocyanate, sodium, and potassium95. Additionally, several protein biomarkers, such as MMP-8, CRP, TNF-α, and IL-6, have shown utility in diagnosing and monitoring periodontal diseases25. In this section, we review continuous biochemical sensors designed for biomarker monitoring in dental and oral health applications. To evaluate and compare the analytical and device performance, we will assess dynamic range, sensitivity, specificity, temporal resolution, and longevity as key metrics for assay development. Table 2 summarizes and compares the principal types of bioanalytical sensors for dental, oral, and craniofacial applications.
a Overview of biochemical sensor design, components, and working principles. b The target recognition elements, such as antibodies221, aptamers138, enzymes10, and ion-specific material156 can specifically interact with the analytes through binding/enzymatic reactions. c The signal transduction module converts the molecular reaction events into a signal that can be measured, such as electrical and optical signals138,139,140. d Examples of sensor platforms for oral applications include pacifiers16, mouthguards11,164, and tooth‑mounted devices8.
Cytokine monitoring
Cytokines, including IL-1b, IL-6, IL-8, IL-17, and TNF-α, are important biomarkers for oral disease such as gingivitis, periodontitis, and oral cancers96,97,98,99,100,101. Previous studies have shown that concentrations of IL-6, IL-8, and IL-17 are elevated in patients with chronic periodontitis compared to periodontally healthy individuals96,102. For OSCC, salivary levels of IL-6 and IL-8 are significantly higher in patients compared to control populations97,103. Another study reports that severe gingivitis patients exhibit significantly elevated salivary IL-6, IL-1β, and CRP compared to controls104. Gingival crevicular fluid and salivary cytokine concentrations are summarized in Table 3. Alterations in cytokine biomarkers reflect disease status and progression. Precise quantification enables dentists to intervene before the disease advances to a severe stage.
Recent studies have explored continuous biochemical sensors capable of monitoring the aforementioned pro-inflammatory cytokines in biofluids, including saliva83,85,87,88,89,90,91,92. While a few wearable sensors have been designed to continuously track cytokines84,86 in sweat and other peripheral biofluids, no wearable platforms have yet been successfully implemented for continuous cytokine detection in the oral cavity. Nonetheless, several existing cytokine biosensors exhibit design features and performance specifications that could be engineered and adapted for intraoral use. Cytokine biosensors primarily employ antibodies87,88,89,90,91,92 or aptamers83,84,85,86 as their biorecognition elements. Another biosensor employs complementary oligonucleotides to quantify IL-8 mRNA92. These sensors typically transduce signals via electrochemical83,85,86,87 or optical89,91 methods. Electrochemical cytokine sensors employ potentiometric transducers, such as field-effect transistors85,87 (Fig. 3a–c), to measure electrode potential differences, and amperometric transducers to quantify reaction currents at a fixed potential83,86. Electrochemical transduction enables facile miniaturization and provides biocompatible, stable electrode interfaces. Optical cytokine biosensors, such as SPR platforms in a study89,91, detect IL-8 by monitoring resonance shifts when the analyte binds to an anti-IL-8 coated surface. Optical biosensors typically depend on bulky instrumentation, hindering their miniaturization into intraoral wearable formats. No intraoral wearable cytokine biosensor has been reported, although promising formats for cytokine detection in other biofluids have been proposed. A recent study integrated electrochemical fibrous sensors into wristband and headband formats to monitor IL-6 in sweat86 (Fig. 3d–f). Another implanted aptamer-based sensor employing oscillation-driven active reset and electrochemical transduction quantified IL-6 and TNF-α in interstitial fluid84. Wearable cytokine sensors have demonstrated effective recognition, signal transduction, and operational durability in non-oral contexts, indicating their potential for adaptation to intraoral formats. An IL-6 sensor was created by modifying a nanochannel film with a catalyst and antibodies. The sensor was built on an ITO electrode using a straightforward growth method. Its surface was functionalized to attach capture antibodies, creating a specific detection interface for IL-6 under neutral conditions. IL-6 binding to this interface changed the sensor’s electrical signal, enabling precise measurement of IL-6 levels within 1 fg/mL to 10 ng/mL105 (Fig. 3g, h).
a An aptamer-based field effect transistor biosensors responds to the change of IL-6 concentration within 400 s in saliva with a detection range from 50 pm to 100 nM (b) for 80 mins in saliva (c)85. d Another novel electrochemical fabric based on aptamer-functionalized carbon nanotube/graphene fibers could detect IL-6 in a range from 1 pg/ml to 100 ng/ml (e) for 3 h (f)86. g An IL-6 sensor demonstrated continuous monitoring from 1 fg/mL to 10 ng/mL for 60 min in GCF (h)105.
Most electrochemical and optical cytokine biosensors achieve detection limits from low pg/mL to ng/mL, encompassing physiological cytokine concentrations. By employing target-specific aptamers or antibodies, these cytokine sensors achieve high specificity. Nearly all continuous cytokine biosensors yield only rising signals, as slow target dissociation and the absence of active reset mechanisms prevent real-time tracking of dynamic concentration changes84. Table 4 summarizes the performance metrics of the cytokine sensors. Interestingly, an electrochemiluminescence (ECL) biosensor for IL-6 achieved attomolar-to-picomolar sensitivity in gingival crevicular fluid, but its reliance on bulky instrumentation prevents miniaturization for wearable oral applications105. Several continuous cytokine biosensors have demonstrated accurate target monitoring over multi-hour durations85,86,89. In one study, the wearable sweat-based IL-6 sensor retained performance after roughly 1000 use cycles without significant degradation86. Continuous cytokine tracking has been realized in saliva and gingival crevicular fluid in non-intraoral setting using electrochemical and optical biosensor platforms. To date, no intraoral wearable has been reported that can capture cytokines in real time.
Future advancements will demand miniature, robust electrochemical or optical transducers designed for the oral cavity, biocompatible anti-fouling coatings to reduce saliva-induced drift, and low-power wireless telemetry for uninterrupted data acquisition.
Quantifying other protein analytes
Non-cytokine salivary and GCF proteins such as MMPs, ALP, Osteoprotegerin (OPG), and C-reactive protein (CRP) also serve as key oral health biomarkers106,107,108,109,110 (Table 5). Levels of enzymes such as MMPs (e.g., MMP-8 and MMP-9) and ALP are significantly elevated in GCF and saliva of chronic periodontitis patients compared to healthy individuals111,112,113,114. Elevated levels of MMPs, particularly MMP-2, MMP-9, and MMP-12, are frequently observed in oral cancer and are associated with tumor progression and metastasis115,116. OPG is markedly lower in chronic periodontitis than in healthy individuals117, whereas CRP levels increase stepwise from health to gingivitis to periodontitis118. Besides, children with reduced salivary α-amylase (sAA) activity exhibit increased susceptibility to dental caries119. Timely detection of non-cytokine protein biomarkers informs oral disease severity and progression, enabling early intervention.
Many biosensors have been developed to quantify non-cytokine salivary and GCF proteins implicated in oral and dental health120,121,122,123,124,125,126,127,128,129. Most current biosensors are configured for single measurements, although certain designs could be adapted for continuous monitoring and wearable integration. Non-cytokine protein biosensors rely on antibody-based affinity recognition123,124,125,126,128,129 or enzymatic reaction-based recognition122,127 mechanisms. Electrochemical transduction predominates among these biosensors, with optical and acoustic methods also employed for signal transduction.
In an rGO-based electrochemical sensor, MMP-7 cleaves surface-bound peptides, reducing their mass and charge and thus lowering the measured current127. In another ALP sensor, ALP catalyzes phenyl phosphate hydrolysis to phenol, which is quantified by square-wave voltammetry122. In another example, an ultrasensitive electrochemical immunoassay achieved rapid, simultaneous IL-6 and MMP-9 detection on a heated screen-printed carbon electrode (HSPCE) modified with hydrophilic, low-toxicity graphene nanoribbons for antibody immobilization and signal amplification. PS@PDA-metal nanocomposites labeled the detection antibodies, producing a strong voltametric response in acetic buffer. A sandwich format on adjacent HSPCE electrodes eliminated crosstalk and enabled quantification from 10⁻⁵ to 10³ ng/mL124.
These sensors span hundreds of pg/mL to hundreds of ng/mL, matching salivary and GCF protein levels observed in oral disease. Specificity is achieved using target-specific antibodies or substrate-selective enzymatic reactions. The enzymatic reaction-based biosensors are unsuited for continuous operation because substrate turnover depletes surface-bound reagents and demands external reagent supply, precluding uninterrupted monitoring. Affinity-based electrochemical protein biosensors preserve surface-bound biorecognition elements after each assay, enabling continuous monitoring and integration into wearable formats. Extending these biosensors to continuous operation requires enhanced longevity; applying advanced surface passivation can mitigate degradation and enable prolonged monitoring94,130.
Identification of infection
Oral bacteria critically influence dental and periodontal health by driving biofilm formation, acid production, and inflammatory responses131,132,133,134,135. Clinical studies reveal that S. mutans predominates in individuals with dental caries, while Streptococcus sanguinis levels decline131,136. P. gingivalis abundance is markedly elevated in periodontitis patients(105–108 CFU/mL) compared to healthy individuals137. Other bacteria/pathogen such as Actinomyces species, Fusobacterium nucleatum, and T. denticola—contribute to plaque maturation and tissue destruction. These microbial-level changes highlight the need for biosensors capable of sensitive, species-specific detection in saliva or crevicular fluid. Such devices must discriminate closely related bacteria, operate across clinically relevant concentration ranges, and ideally support multiplexed, real-time monitoring to enable early diagnosis and guide personalized oral care.
Continuous and end-point biosensors have been developed for salivary bacteria and pathogen detection93,138,139,140,141,142,143, and some wearable devices enable intraoral microbial monitoring15. Bacterial biosensors employ affinity-based recognition elements such as aptamers138, antibodies141,142, antimicrobial peptide (AMP)15,93, and molecularly imprinted polymers (MIP)144 or an indirect detection method that monitors bacteria-mediated reactions143. These bacterial biosensors predominantly employ electrochemical transduction. A biosensor integrates an S. mutans specific DNA aptamer and MIP on an electrode, using methylene blue as a redox probe. Binding of S. mutans occludes electron transfer between methylene blue and the electrode, causing a current decrease proportional to bacterial concentration. It quantifies S. mutans across the clinically relevant range for high caries risk and stable for about 60 days138 (Fig. 4a–c). A wash-free electrochemical assay for P. gingivalis uses glycine-proline-arginine-4-aminophenol (with glycylglycine boosting Arg-gingipain activity) as the proteolytic substrate. Released 4-aminophenol undergoes EC redox cycling with tris(2-carboxyethyl)phosphine, and matrix-corrected charge differences before and after a 15-min incubation yield high, reproducible, pathogen-specific signals in saliva139 (Fig. 4d–f). Indirect assays requiring exogenous substrates preclude continuous or wearable implementation due to the need for reagent replenishment. A graphene sensor functionalized with self-assembled antimicrobial peptides enables bio-selective, single-cell bacterial detection and is powered wirelessly via an integrated resonant coil, eliminating onboard batteries or wiring. Printed on water-soluble silk, it can be bio-transferred onto tooth enamel for seamless intraoral integration. It demonstrated bacterial detection in seconds across at least five consecutive cycles15. By interchanging the surface affinity element, the platform can be tailored to detect S. mutans, S. sanguinis, or P. gingivalis in dental applications.
a A portable saliva sensor based on aptamer and S. mutans-imprinted polymers (SIPs) demonstrated measurement of S. mutans from 103–109 CFU/mL (b) and is stable for 60 days (c)138. d A washing- and separation-free electrochemical sensor demonstrated indirect detection of P. Gingivalis in saliva from a range 105–108 CFU/mL e, f139.
Reported bacterial biosensors typically span 102–109 CFU/mL, covering physiological salivary pathogen concentrations15,138,141,142. Aptamer- and antibody-based sensors achieve high pathogen specificity, whereas antimicrobial peptide–based sensors lack species-level discrimination due to semi-selective binding of AMP15. Performance metrics for these biosensors are summarized in Table 6.
Biosensor for ions
Ion and metabolite concentrations in saliva and gingival crevicular fluid regulate enamel mineralization, microbial balance, and inflammatory responses, making them critical oral health biomarkers145,146. Clinical studies have shown that periodontitis patients exhibit higher salivary sodium, potassium, calcium, and uric acid than healthy individuals147,148,149. In caries prevention, maintaining salivary fluoride150,151,152 within a therapeutic window and adequate calcium153 and phosphate154 levels supports enamel repair. Altered trace elements—copper, zinc, lead, cadmium, calcium, and magnesium—have been associated with oral potentially malignant disorders and OSCC155. These observations underscore the need for biosensors offering selective ion and metabolite detection across physiological ranges, robust performance in complex biofluids, and multiplexed or continuous real-time monitoring.
Multiple biosensors have been reported measuring salivary ions such as sodium16,81,82, potassium16, uric acid10,156, fluoride79, and phosphate157. Wearable sensors are already available for sodium81,82, potassium16, and uric acid10, continuous sensors are available for fluoride79 and phosphate158. Wearable salivary ion sensors predominantly utilize ion selective electrodes16,79,80,81,82, with some designs based on enzymatic or catalytic reactions10,157 and others incorporating ion selective nanomaterials156. Most of these sensors employ electrochemical transduction. An ISE biosensor quantifies Na⁺ or K⁺ activity via a selective membrane that generates a potential, proportional to the logarithm of ion activity, measured against a reference electrode. Its ion-selective membrane also serves as an antifouling barrier, protecting the electrode and maintaining stability for up to 1 week16 (Fig. 5a–d). An enzymatic electrochemical uric acid sensor catalyzes UA oxidation to modulate electron flux at the electrode, generating a current proportional to UA concentration. A phenylenediamine membrane provides antifouling protection and sustains stable performance for up to 4 days10 (Fig. 5e, f). A SERS based salivary UA sensor employs Au/MIL-125(Ti) nanocomposites that selectively adsorb UA via π–π stacking with benzene rings of MIL-125(Ti), enabling rapid, sensitive detection156.
ISE sensors detect sodium and potassium from 10⁻⁴ to 1 M and fluoride from 10⁻⁶ to 10⁻² M79, while the UA sensor spans 0–1000 µM10, fully covering physiological salivary ranges. By using ion selective membranes, nanocomposite adsorption, or enzyme catalyzed reactions, these sensors achieve high specificity for their target ions. However, ISE sensors can exhibit signal crosstalk when non target ions permeate the selective membrane81. These ion sensors provide rapid responses, typically within tens of seconds. Their stability and facile miniaturization enable integration into intraoral wearables such as mouthguards10,11,12,13,79,81, tooth mounted films14,15, and pacifiers16,17. Performance metrics for these biosensors are summarized in Table 7.
Small molecule detection
Salivary glucose and lactate critically influence oral health by modulating immune defenses, biofilm acidity, and tissue integrity. In diabetic periodontitis, salivary glucose rises above physiological levels, impairing leukocyte chemotaxis, thickening the vascular basement membrane, and weakening periodontal defenses159, while also fueling lactic acid production by oral biofilms. Elevated salivary lactate further lowers plaque pH, accelerating enamel demineralization and caries160,161. These pathological elevations—relative to normal saliva glucose (0.5–1.0 mg/100 mL) and lactate (0.1–2.5 mmol/L)—underscore the need for biosensors with micromolar-to-millimolar sensitivity, robust anti-fouling design, and real-time readout capability to detect and track metabolic shifts in saliva162.
Multiple wearable biosensors have been reported measuring salivary glucose11,13,14,17,78 and lactate12,162. Most Glucose and lactate biosensors rely on enzyme-catalyzed recognition coupled with electrochemical transduction. In an enzymatic electrochemical glucose sensor, an electrode immobilized glucose oxidase (GOx) oxidizes glucose to gluconolactone and hydrogen peroxide, and the subsequent electrooxidation of hydrogen peroxide at the electrode produces a current proportional to glucose concentration11,13,17,78 (Fig. 6). A mouthguard lactate sensor immobilizes lactate oxidase on the working electrode to catalyze lactate oxidation, generating an electrochemical current proportional to lactate concentration12. In another study, a materials-based strategy embeds an active sensing layer between two reverse-facing split-ring resonators in a compact RF construct, enabling glucose detection via shifts in resonant frequency and amplitude; adding tailored functional coatings could impart molecular specificity. Although this method lacks molecular specificity, tailored functional coatings could be introduced to enable selective analyte binding14.
Enzymatic electrochemical glucose sensors operate over 1–10,000 μM and lactate sensors over 0.1–1.0 mM, covering physiological salivary concentrations. Specificity arises from enzyme–substrate recognition, with immobilized enzymes remaining active for repeated measurements. These electrochemical sensors respond within seconds and retain functional stability for over 30 min. Electrodes are miniaturized and coated with a poly-ortho-phenylenediamine antifouling film, then integrated into wearable platforms such as pacifiers17, mouthguards11,12,13, and tooth-mounts14. The performance metrics for these biosensors are summarized in Table 8.
Biosensor for pH
The pH homeostasis within the body can impact metabolic reactions as well as biological transport systems, which can manifest within the oral cavity163. In normal situations, human pH levels range from 6.6 to 7.1, and salivary pH levels that deviate from the norm are indicative of physiological diseases164,165. Low pH levels of one’s oral microbiome indicate a strong relationship between increased caries risk, enamel demineralization, and gingival inflammation166. pH levels in the oral cavity can also affect the quality and strength of dentures in a patient’s mouth. Decreased oral pH may be caused by everyday activities such as sugar intake, which promotes acid production and bacterial accumulation, or health conditions such as gastroesophageal reflux167. Degradation of chemical structures within the denture material can be amplified in an acidic environment, leading to a rough denture surface, cracks, and inability to resist fractures167. Examining pH levels in the human body can provide insights into occurrences in the oral cavity and digestive tract. These insights can help the provider make informed decisions when choosing the most appropriate denture material for each patient, as well as monitoring their oral health status.
Several electrochemical-based salivary pH sensors have been reported, utilizing diverse transduction principles. A potentiometric pH sensor employing a thin antimony electrode interacts with hydrogen ions to generate a potential proportional to pH, maintaining accurate measurements over pH 1–9 for 24 h. A conductimetric pH sensor employs a polyaniline membrane whose protonation and deprotonation modulate its conductivity, with pH inferred from the resulting resistance changes168. A mouthguard-based voltammetric pH sensor uses proton-selective redox probes anchored to multiwalled carbon nanotubes to convert pH shifts from 2 to 11 into electrical signals169. Current pH sensors are highly mature that deliver rapid (second-scale) responses across pH 2–11, encompassing the full salivary range, and maintain stable performance for over 20 h.
Sensor for dental force measurement
Continuous monitoring of forces is essential for delivering personalized and biologically optimized orthodontic treatment170. A major technical challenge is to capture complex, dynamic forces acting on teeth in vivo, in real-time, and with high precision. Traditional approaches, including finite element simulations or force gauges on brackets, fail to reflect the temporal and spatial variability of intraoral forces, particularly with clear aligners or removable appliances171. To bridge this gap, sensor-integrated systems capable of measuring multidirectional forces continuously in the oral cavity are needed. Typical orthodontic forces vary depending on the type of tooth movement. Tipping and rotational movements generally require 35–60 g (0.343–0.686 N), while bodily movement demands higher forces around 70–120 g (0.686–1.176 N)172. More delicate movements like intrusion use as little as 10–20 g (0.098–0.196 N) to avoid root damage. In clear aligner treatments, the average force per tooth typically falls between 20 and 100 g (0.196–0.98 N)173, though this may decay over time due to material relaxation and intraoral conditions. These ranges provide approximate benchmarks for the sensitivity and resolution in force sensing systems.
Force sensor format and components
Intraoral force sensors typically consist of three core elements: a transducer, a signal processing circuit, and a mechanical interface tailored to the dental anatomy. The transducer converts mechanical forces into readable electrical signals. Typical transducer modalities include piezoresistive, capacitive, or piezoelectric (Fig. 7). These signals are subsequently amplified, filtered, and digitized by a compact electronic device. The mechanical interface packages the force transducer and ensures that intraoral forces are efficiently transferred to the sensing element without compromising patient comfort174. To evaluate sensor performance, both analytical and device characteristics must be considered. Analytical performance includes sensitivity, linearity, repeatability, and response time. Sensitivity in orthodontic force sensing typically falls in the range of 20–50 mV/N for piezoresistive sensors, and 5–15% signal change per newton for capacitive sensors, depending on dielectric design175. Most oral force sensing systems aim for a minimum detectable force between 10 and 50 mN, which is sufficient to capture subtle differences during orthodontic adjustment or bite evaluation176. To ensure accuracy across a broad force range, force sensors often have linearity above 95%, meaning the sensor’s output remains within 5% deviation from an ideal linear response over the entire measurement range, and maintains low hysteresis (<5% of the full-scale output), ensuring consistent signal behavior during loading and unloading cycles177. Repeatability is typically reflected in variations below 3% across measurements, and low signal drift with less than 1% per day, which are essential for long-term monitoring in clinical settings. Fast response times, often under 0.5–1 s, are especially important for capturing transient bite forces or real-time orthodontic changes55.
a Piezoresistive sensor detects force by measuring the change in electrical resistance of a material when it is mechanically strained. b Capacitive sensor measures force based on changes in capacitance caused by the relative displacement of conductive plates separated by a dielectric layer. c Piezoelectric sensor converts mechanical stress into electrical charge using materials that generate voltage when deformed.
Piezoresistive force sensor
Piezoresistive force sensors convert mechanical stress into electrical signals via changes in the resistivity of semiconducting or metal elements. The basic principle relies on strain-induced resistance modulation, typically captured using a Wheatstone bridge configuration178 (Fig. 7a). This mechanism offers high sensitivity (e.g., 37.79 mV V−1 MPa−1)179 in a compact form factor, making it one of the most widely used modalities in intraoral force sensing180.
Several force sensors targeting dental applications have been developed and characterized. Lapatki et al.181 developed a “smart bracket” integrating diffused piezoresistors on a silicon die, achieving a force measurement range of ±1.5 N and moment range of ±15 N·mm, with an accuracy of ±0.04 N in force measurement and ±1 N·mm in moment measurement under laboratory conditions. Shi et al. introduced a rosette-type piezoresistive sensor designed for insertion between clear aligners and tooth surfaces182. This configuration allows temperature-compensated measurement of the in-plane normal stress difference and the shear stress. Their design featured a 100 μm-thin silicon die and reconstructed forces with a resolution of better than 0.05 N. Minor misalignments or non-uniform tray compression in the design can introduce error margins of up to ±0.04 N, which requires in situ calibration. The system showed excellent linearity and demonstrated clear force stage progression profiles throughout a 7-day aligner wear period. Similar sensor for integration with a clear aligner was also reported by Liu et al.183. This sensor was used to quantify the forces of varying thicknesses and activation levels on a resin tooth model. The measured forces were validated against finite element simulation results, showing a standard deviation of 0.2 N (Fig. 8a–c).
a–c Force measurement setup in a thin film piezoresistive force sensor183. b Stress distribution and c the measured force changes over time. d Measured orthodontic force profile over 80 min in an RF-harvested piezoresistive platform55. e Correlation of applied force and device readout. f Measured force response simulated by finger press.
Longevity and drift performance are crucial in prolonged intraoral deployment. Li et al. tested a wireless, RF-harvested piezoresistive platform continuously submerged in 37 °C saline over 6 weeks. The sensor-maintained drift within ±3% and sensitivity of approximately 0.13 N across the 0–2 N range (Fig. 8d–f). This study was one of the first to simulate both the biochemical and thermal conditions of the oral cavity over extended durations, providing evidence that piezoresistive sensors can achieve multi-week operational stability without recalibration or rebonding55.
Capacitive force sensor
Capacitive force sensors operate on the principle that capacitance changes when the applied load perturbs the plate separation, effective area, or the permittivity of the dielectric medium, thus changing the voltage in the bridge circuit composed of capacitors and other resistive elements. (Fig. 7b) Compared to resistive gauges, capacitive sensors have reduced signal noise and better repeatability184, highlighting capacitive sensing’s potential to enhance precision in real-time orthodontic monitoring.
Capacitive force sensors have been incorporated in orthodontic force measurements. Hafner et al. integrated four sensors beneath a 0.018” × 0.025” bracket slot and achieved accurate reconstruction of full 3D force (±0.05 N) and moment vectors (±100 N mm)185. To improve biocompatibility in the oral environment, Sun et al. present a novel superhydrophobic capacitive sensor with a detection range of up to 400 N186. By leveraging a porous and superhydrophobic architecture, the sensor achieves a high sensitivity of 3.2% signal change per kPa, excellent durability (over 15,000 cycle tests), and effective mitigation of biofouling issues186. Ogihara et al. developed a sensing device capable of measuring spatially-resolved force, enabling the quantification of pediatric bite force on individual teeth187 (Fig. 9a–c). Wang et al. designed an ultra-thin capacitive sensor tailored for clear aligner systems, which use an inductive coil as the transmission unit, a flat capacitor as the signal transducer, and resonance frequency as the readout signal174. The sensor exhibited a rapid response time under simulated dynamic conditions (<0.5 s), excellent hysteresis and stability over time174 (Fig. 9d–f).
a The sensor using a capacitive surface pressure distribution187. b, c Calculated results of bite forces for the first and second primary mola, these two calculations suggest that the newly developed capacitive surface pressure distribution sensor can measure occlusal pressure in the first permanent and second primary molars with good reproducibility187. d Sensor hysteresis174. e System response time and recovery time174. f Sensor repeatability, the Variance is 0.27174.
Piezoelectric force sensor
When a piezoelectric material is mechanically deformed (compressed, stretched, or sheared), it produces an electric potential across its surfaces that is proportional to the applied force and can be measured as a voltage signal188 (Fig. 7c). This type of transducer is highly sensitive and has a fast response time. It is best suited for transient forces measurement, such as bite forces189, but has limited responsiveness to low-frequency or static loads, such as orthodontic treatments190.
An early example of a piezoelectric force transducer in oral application integrates a PVDF-based piezoelectric aligner that sensed forces and also actuated at 30 Hz for potential therapeutic modulation191,192. This system differentiated bite and chewing events in the 0–2 N range, with repeatable transduction over multiple cycles. More recently, Hu et al. developed a six-dimensional piezoelectric sensor based on a tenon-and-mortise structure, capable of resolving both force (±1 N) and moment (±4 N mm) across six axes193 Their sensor demonstrated multiaxial sensitivity and low hysteresis in simulated orthodontic loading, offering a comprehensive force vector profile that surpasses uniaxial or planar-only devices. For device integration towards clinical use, Feng et al. reported a high-density piezoelectric fiber array into a transparent aligner for real-time monitoring of bite forces and parafunctional activity194 (Fig. 10). The highly integrated device in an aligner allows it to monitor bad oral habits, such as lip biting (LB), thumb sucking (TS), and grinding teeth (GT), in addition to detecting the bite status of teeth, as shown in Fig. 10d.
a Schematic illustration of ARIA showing the piezoelectric sensor array positioned on the occlusal surface of the teeth, along with the printed circuit board and battery. The exploded view of the board (bottom left) highlights the components, including the analog front-end, analog-to-digital converter, and microcontroller unit. b Confusion matrix showing the accuracy of the machine learning model in predicting the malocclusion in the test set. c Structural scheme of a 6-layer composite flexible piezoelectric sensor. d Photographs of a subject wearing ARIA and performing CO (centric occlusion), LB (lip biting), TS (thumb sucking), and GT (grinding teeth), and the corresponding voltage curve generated during these activities.
Clinical applications of force sensors
Unlike static assessments using wax bites or tension gauges, embedded sensors allow dynamic monitoring of force magnitude, direction, and duration in vivo, enabling personalized treatment, early error detection, and remote therapeutic feedback. Their applications broadly fall into two major categories: orthodontic force measurement, which aims to optimize tooth movement by quantifying the forces delivered by brackets or aligners, and bite force measurement, which helps evaluate occlusal function and assess masticatory efficiency in prosthetic and surgical rehabilitation contexts. The clinical utility of these sensors is enhanced by their ability to operate wirelessly and continuously in the complex oral environment. By integrating into aligners, brackets, or occlusal splints, these miniaturized platforms provide clinicians with actionable clinical data that support clinical decision-making.
Orthodontic force measurement
Accurately quantify and continuously monitor the three-dimensional loading and temporal adaptation of forces on teeth are of great significance. Studies show that periodontal tissues undergo their most pronounced stress-relaxation within the first 48 h195,196 and then level off over the next 7–14 days196,197; accordingly, clear aligners themselves are typically replaced every 7–14 days198. To achieve continuous force monitoring during this period, several factors such as power consumption, device size and weight, and device robustness in the oral environment need to be considered.
Traditional on-board battery solutions for wearable or implantable systems face limitations in size, longevity, and maintenance. Recent oral biosensor developments have shown energy-efficient architectures and passive communication. Kuhl et al. introduced a battery-free sensor that harvested power via a 13.56 MHz NFC link and streamed data wirelessly at 10 Hz199. The device was successfully demonstrated during simulated canine retraction. Similarly, Li et al. reported a piezoresistive platform that harvested RF energy and logged orthodontic forces for over 6 weeks in 37 °C saline, which is enough to span a full wire adjustment interval without requiring sensor replacement or rebonding55.
For long-term clinical use, intraoral sensors must be both flexible and miniaturized to ensure comfort and preserve normal oral function. To address this, Lapatki et al. developed a “smart bracket” embedding 32 piezoresistors within a standard twin-bracket footprint, maintaining compatibility with conventional bonding protocols181. Kuhl’s subsequent CMOS integration on a 2.9 × 2.9 mm die further pushed miniaturization by embedding 24 stress sensors and an ADC array into a single compact system suitable for clinical integration with brackets200. Shi et al. addressed this by back-thinning silicon to just 100 µm, enabling the placement of a rosette-pattern piezoresistive chip between an aligner surface and tooth182. The device preserved aligner conformity while enabling accurate 3D force reconstruction. Wang et al. built on this approach by creating an ultra-thin, flexible capacitive film sensor for aligners that provides wireless, real-time, and quantitative force measurements with high positional specificity and sensitivity, supporting clinical comparisons and early detection of staging errors174.
The intraoral conditions are characterized by moisture, fluctuating pH, and continuous mechanical stress, which degrade sensor performance over time. To achieve sensor durability in this environment, packaging strategies have evolved from simple silicone coatings to fully hermetic encapsulations using biocompatible polymers. Some systems have demonstrated multi-week operation without signal drift, validating their ability to withstand harsh intraoral conditions55.
Bite force measurement
A fundamental requirement in bite force sensing is the ability to accommodate the wide range of occlusal forces encountered in vivo. In healthy adults, molar forces typically range from 300 to 600 N60, with peak loads exceeding 800 N201,202,203. This places stringent durability demands on sensors, particularly their ability to endure repetitive high-impact compression without calibration drift or mechanical failure. To address this, traditional strain gauge-based devices often utilize metallic forks coated in resilient polymers to buffer occlusal loads and extend device lifespan. However, such rigid designs204,205 often causes discomfort and alter natural biting behavior, compromising both patient compliance and reliability for long-term use. To support continuous force monitoring, there is a growing need for flexible, conformable sensors that can withstand the harsh intraoral environment while preserving normal oral function206,207. Pressure-based bite transducers using pneumatic scaffolds and fluid-filled deformable chambers offer enhanced adaptability by more evenly distributing occlusal forces across curved dental arches58,208. Unlike orthodontic force sensors, which are confined to research prototypes, bite force sensors have been deployed in population-level studies evaluating correlations between craniofacial morphology and bite strength209,210. However, limited dynamic range and response lag hinder its use in real-time diagnostics or time-resolved studies of mastication dynamics. To overcome these limitations, next-generation systems have incorporated ultra-thin piezoelectric film sensors (~0.1 mm), offering both high spatial and temporal resolution211,212.
Beyond functional bite, bruxism concentrates on the same engineering requirements: nocturnal events are brief, clustered, and can include peaks that approach or exceed daytime voluntary effort213. Clinically, recent cohort work links bruxism with higher maximum bite force and greater rates of tooth or restoration fractures, and calls for bite force methods to guide prevention and treatment, strengthening the case for instrumented force monitoring214. Based on these engineering requirements for bruxism, the bite force sensor designed for the application of bruxism emphasizes these performance indicators: dynamic range and impact robustness, form factor and comfort, and responsibility. To satisfy these constraints in practice, mouthguard and splint carriers have become the dominant form215. Pressure-sensitive polymer stacks with on-board thresholding and time stamping support multi-night episode logging at low average power, whereas ultrathin electret and dielectric-elastomer films add self-biased or self-powered sensing in submillimeter laminates for prolonged wear216. And flexible PVDF films offer high sampling rates and the option of small arrays to map where loads concentrate217.
Conclusions and future directions
Recent advances in biosensor technology have demonstrated substantial potential for detecting a broad spectrum of oral biomarkers, including ions (e.g., sodium, potassium, fluoride), small molecules (e.g., glucose, lactate), pH, microbial species, and proteins. These analytes are instrumental in deepening our understanding of oral biology and enhancing the early diagnosis, monitoring, and management of oral diseases. While many biosensors have shown robust analytical performance and long-term stability in vitro, their integration into clinical settings remains a critical hurdle.
Emerging platforms developed for other biofluids, such as sweat, interstitial fluid, and gastrointestinal secretions, offer valuable insights and design strategies that may be adapted for intraoral applications. However, the oral cavity presents unique challenges, including a highly dynamic biochemical environment influenced by diet and hygiene routines, constant mechanical motion, and broad temporal and physiological variability. These conditions demand bio-interfaces that are not only highly sensitive and selective but also mechanically durable, biocompatible, and minimally obtrusive.
Most current efforts in oral biosensing have focused on developing proof-of-concept devices, with limited demonstrations of clinical utility or integration into standard care workflows. A more integrated research and development effort is needed to address commercialization and regulatory barriers that hinder translation. On the commercialization side, sensor designs that not only optimize analytical performance, but also consider scalable manufacturing processes and optimize user acceptance, comfort, and long-term adherence are needed. On the regulatory side, pathways for intraoral biosensors in constant contact with the oral mucosal tissues or saliva are still emerging. Demonstrating long-term safety, reliability, and data integrity will be critical for approval and adoption. A key next step will be the systematic generation of high-quality, clinically relevant data that can validate the impact of continuous, real-time oral biomarker monitoring on diagnosis, treatment, and patient outcomes. Ultimately, bridging the gap between technological innovation and clinical translation will be essential to fully realize the promise of biosensing in advancing precision oral healthcare.
Data availability
No datasets were generated or analysed during the current study.
References
-
Yactayo-Alburquerque, M. T., Alen-Méndez, M. L., Azañedo, D., Comandé, D. & Hernández-Vásquez, A. Impact of oral diseases on oral health-related quality of life: a systematic review of studies conducted in latin america and the caribbean. PLoS ONE 16, e0252578 (2021).
-
Wang, K.-N. et al. The applications of flexible electronics in dental, oral, and craniofacial medicine. Npj Flex. Electron. 8, 33 (2024).
-
Fellows, J. L., Atchison, K. A., Chaffin, J., Chávez, E. M. & Tinanoff, N. Oral health in America. J. Am. Dent. Assoc. 153, 601–609 (2022).
-
De Magalhães, A. A. & Santos, A. T. Advancements in diagnostic methods and imaging technologies in dentistry: a literature review of emerging approaches. J. Clin. Med. 14, 1277 (2025).
-
Coachman, C., Bohner, L., Jreige, C. S., Sesma, N. & Calamita, M. Interdisciplinary guided dentistry, digital quality control, and the “ copy-paste ” concepts. J. Esthet. Restor. Dent. 33, 982–991 (2021).
-
Moulahoum, H. & Ghorbanizamani, F. Unexploited opportunities in oral disease biosensors and digital health integration. Clin. Chim. Acta 576, 120401 (2025).
-
Sharma, A., Badea, M., Tiwari, S. & Marty, J. L. Wearable biosensors: an alternative and practical approach in healthcare and disease monitoring. Molecules 26, 748 (2021).
-
Shi, Z. et al. Wearable battery-free theranostic dental patch for wireless intraoral sensing and drug delivery. Npj Flex. Electron. 6, 49 (2022).
-
Liu, Y., Yue, W. & Cui, Y. Development of an amperometric biosensor on a toothbrush for glucose. Sens. Actuators Rep. 5, 100133 (2023).
-
Kim, J. et al. Wearable salivary uric acid mouthguard biosensor with integrated wireless electronics. Biosens. Bioelectron. 74, 1061–1068 (2015).
-
Arakawa, T. et al. Mouthguard biosensor with telemetry system for monitoring of saliva glucose: a novel cavitas sensor. Biosens. Bioelectron. 84, 106–111 (2016).
-
Kim, J. et al. Non-invasive mouthguard biosensor for continuous salivary monitoring of metabolites. Analyst 139, 1632–1636 (2014).
-
Arakawa, T. et al. A wearable cellulose acetate-coated mouthguard biosensor for in vivo salivary glucose measurement. Anal. Chem. 92, 12201–12207 (2020).
-
Tseng, P., Napier, B., Garbarini, L., Kaplan, D. L. & Omenetto, F. G. Functional, RF-trilayer sensors for tooth-mounted, wireless monitoring of the oral cavity and food consumption. Adv. Mater. 30, 1703257 (2018).
-
Mannoor, M. S. et al. Graphene-based wireless bacteria detection on tooth enamel. Nat. Commun. 3, 763 (2012).
-
Lim, H.-R. et al. Smart bioelectronic pacifier for real-time continuous monitoring of salivary electrolytes. Biosens. Bioelectron. 210, 114329 (2022).
-
García-Carmona, L. et al. Pacifier biosensor: toward noninvasive saliva biomarker monitoring. Anal. Chem. 91, 13883–13891 (2019).
-
Khaleque, M. A. et al. Nanostructured wearable electrochemical and biosensor towards healthcare management: a review. RSC Adv. 13, 22973–22997 (2023).
-
Ajami, S. & Teimouri, F. Features and application of wearable biosensors in medical care. J. Res. Med. Sci. 20, 1208 (2015).
-
Baker, J. L., Mark Welch, J. L., Kauffman, K. M., McLean, J. S. & He, X. The oral microbiome: diversity, biogeography and human health. Nat. Rev. Microbiol. 22, 89–104 (2024).
-
Larvin, H., Kang, J., Aggarwal, V. R., Pavitt, S. & Wu, J. Risk of incident cardiovascular disease in people with periodontal disease: a systematic review and meta-analysis. Clin. Exp. Dent. Res. 7, 109–122 (2021).
-
Li, Y. et al. Association between periodontitis and dental caries: a systematic review and meta-analysis. Clin. Oral Investig. 28, 306 (2024).
-
Nagdeve, S. N., Suganthan, B. & Ramasamy, R. P. Perspectives on the application of biosensors for the early detection of oral cancer. Sensors 25, 1459 (2025).
-
Gurenlian, J. R. The role of dental plaque biofilm in oral health. Am. Dent. Hyg. Assoc. 81, 116–116 (2007).
-
Dogra, S., Dhawan, P., Tomar, S. S., Kakar, A. & Kakar, E. Intraoral biosensors: a promising diagnostic tool. Curr. Med. Res. Pract. 12, 118 (2022).
-
Lohcharoenkal, W., Abbas, Z. & Rojanasakul, Y. Advances in nanotechnology-based biosensing of immunoregulatory cytokines. Biosensors 11, 364 (2021).
-
Steigmann, L. et al. Biosensor and lab-on-a-chip biomarker-identifying technologies for oral and periodontal diseases. Front. Pharmacol. 11, 588480 (2020).
-
Huang, D. et al. Association between periodontal disease and systemic diseases: a cross-sectional analysis of current evidence. Mil. Med. Res. 11, 74 (2024).
-
Hajishengallis, G. & Chavakis, T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat. Rev. Immunol. 21, 426–440 (2021).
-
Hooshiar, M. H. et al. Recent advances in nanomaterial-based biosensor for periodontitis detection. J. Biol. Eng. 18, 28 (2024).
-
Bodet, C., Chandad, F. & Grenier, D. Potentiel pathogénique de porphyromonas gingivalis, treponema denticola et tannerella forsythia, le complexe bactérien rouge associé à la parodontite. Pathol. Biol. 55, 154–162 (2007).
-
Shaddox, L. M. & Walker, C. B. Treating chronic periodontitis: current status, challenges, and future directions. Clin. Cosmet. Investig. Dent. 2, 79–91 (2010).
-
Dabra, S., China, K. & Kaushik, A. Salivary enzymes as diagnostic markers for detection of gingival/periodontal disease and their correlation with the severity of the disease. J. Indian Soc. Periodontol. 16, 358–364 (2012).
-
Luchian, I., Goriuc, A., Sandu, D. & Covasa, M. The role of matrix metalloproteinases (MMP-8, MMP-9, MMP-13) in periodontal and peri-implant pathological processes. Int. J. Mol. Sci. 23, 1806 (2022).
-
Neurath, N. & Kesting, M. Cytokines in gingivitis and periodontitis: from pathogenesis to therapeutic targets. Front. Immunol. 15, 1435054 (2024).
-
Roberts, H. M., Yonel, Z., Kantarci, A., Grant, M. M. & Chapple, I. L. C. Impact of gingivitis on circulating neutrophil reactivity and gingival crevicular fluid inflammatory proteins. Int. J. Environ. Res. Public. Health 19, 6339 (2022).
-
Takefuji, Y. Dental implant prevalence and durability: a concise review of factors influencing success and failure. Biomater. Biosyst. 17, 100109 (2025).
-
Ting, M. & Suzuki, J. B. Peri-implantitis. Dent. J. 12, 251 (2024).
-
Heitz-Mayfield, L. J. A. Peri-implant mucositis and peri-implantitis: key features and differences. Br. Dent. J. 236, 791–794 (2024).
-
Bernabe, E. et al. Trends in the global, regional, and national burden of oral conditions from 1990 to 2021: a systematic analysis for the global burden of disease study 2021. Lancet 405, 897–910 (2025).
-
Kassebaum, N. J. et al. Global burden of untreated caries: a systematic review and metaregression. J. Dent. Res. 94, 650–658 (2015).
-
Padmanabhan, V. et al. Salivary alpha-amylase activity and its association with early childhood caries and rampant caries experience: a cross-sectional study. Front. Med. 11, 1480139 (2025).
-
Nascimento, M. M., Gordan, V. V., Garvan, C. W., Browngardt, C. M. & Burne, R. A. Correlations of oral bacterial arginine and urea catabolism with caries experience. Oral Microbiol. Immunol. 24, 89–95 (2009).
-
Jiang, Z., Shao, L., Zhou, J., Shao, X. & Shen, C. Causes of death after oral cancer diagnosis: a population based study. Front. Oncol. 14, 1481688 (2024).
-
Akashanand et al. Burden of oral cancer and associated risk factors at national and state levels: a systematic analysis from the global burden of disease in India, 1990–2021. Oral Oncol. 159, 107063 (2024).
-
Silva, L. C. et al. The importance of early treatment of oral squamous cell carcinoma: case report. Oral Oncol. 144, 106442 (2023).
-
Bastías, D., Maturana, A., Marín, C., Martínez, R. & Niklander, S. E. Salivary biomarkers for oral cancer detection: an exploratory systematic review. Int. J. Mol. Sci. 25, 2634 (2024).
-
Singh, P., Verma, J. K. & Singh, J. K. Validation of salivary markers, IL-1β, IL-8 and Lgals3bp for detection of oral squamous cell carcinoma in an indian population. Sci. Rep. 10, 7365 (2020).
-
Oshin, M. et al. Salivary and serum interleukin-6: a credible marker for predicting oral leukoplakia and oral squamous cell carcinoma by enzyme-linked immunosorbent assay (ELISA). Cureus 16, e59113 (2024).
-
Liu, S. et al. Saliva microbiome alterations in dental fluorosis population. J. Oral Microbiol. 15, 2180927 (2023).
-
Thomas, P. A., Shenoy, R. P., Jodalli, P. Pasha, I. & Junaid. Biosensors in the field of dentistry. J. Clin. Diagn. Res. 16, ZE01–ZE04 (2022).
-
Xie, Q. et al. Fabrication of three-dimensional orthodontic force detecting brackets and preliminary clinical test for tooth movement simulation. Heliyon 9, e19852 (2023).
-
He, R. et al. Flexible miniaturized sensor technologies for long-term physiological monitoring. Npj Flex. Electron. 6, 20 (2022).
-
Yashin, D. et al. Markers in blood and saliva for prediction of orthodontically induced inflammatory root resorption: a retrospective case controlled-study. Prog. Orthod. 18, 27 (2017).
-
Li, P. et al. RF energy harvesting for intraoral orthodontic force monitoring. Nano Energy 121, 109244 (2024).
-
Sonnesen, L., Bakke, M., & Beni Bite force in pre-orthodontic children with unilateral crossbite. Eur. J. Orthod 23, 741–749 (2001).
-
Ingervall, B. & Helkimo, E. Masticatory muscle force and facial morphology in man. Arch. Oral Biol. 23, 203–206 (1978).
-
Pereira, L. J., Pastore, M. G., Bonjardim, L. R., CasTELo, P. M. & Gavião, M. B. D. Molar bite force and its correlation with signs of temporomandibular dysfunction in mixed and permanent dentition. J. Oral Rehabil. 34, 759–766 (2007).
-
Rentes, A. M., Gavião, M. B. D. & Amaral, J. R. Bite force determination in children with primary dentition. J. Oral Rehabil. 29, 1174–1180 (2002).
-
Bakke, M. Bite force and occlusion. Semin. Orthod. 12, 120–126 (2006).
-
Sonnesen, L. Malocclusion traits and symptoms and signs of temporomandibular disorders in children with severe malocclusion. Eur. J. Orthod. 20, 543–559 (1998).
-
Marian, D. et al. Challenges and innovations in alveolar bone regeneration: a narrative review on materials, techniques, clinical outcomes, and future directions. Medicina 61, 20 (2024).
-
Rani, S., Bandyopadhyay-Ghosh, S., Ghosh, S. B. & Liu, G. Advances in sensing technologies for monitoring of bone health. Biosensors 10, 42 (2020).
-
Shekhar, A. et al. Salivary biomarkers and temporomandibular disorders: a systematic review. Natl. J. Maxillofac. Surg. 14, 354–359 (2023).
-
Jasim, H., Ghafouri, B., Gerdle, B., Hedenberg-Magnusson, B. & Ernberg, M. Altered levels of salivary and plasma pain related markers in temporomandibular disorders. J. Headache Pain 21, 105 (2020).
-
Alam, M. K. et al. Salivary biomarkers and temporomandibular disorders: a systematic review conducted according to PRISMA guidelines and the Cochrane Handbook for systematic reviews of interventions. J. Oral Rehabil. 51, 416–426 (2024).
-
Karakoulaki, S., Tortopidis, D., Andreadis, D. & Koidis, P. Relationship between sleep bruxism and stress determined by saliva biomarkers. Int. J. Prosthodont. 28, 467–474 (2015).
-
Sáenz-Ravello, G., Hernández, M., Baeza, M. & Hernández-Ríos, P. The role of oral biomarkers in the assessment of noncommunicable diseases. Diagnostics 15, 78 (2024).
-
Fadli, N. A., Abdul Rahman, M., Karsani, S. A. & Ramli, R. Oral and gingival crevicular fluid biomarkers for jawbone turnover diseases: a scoping review. Diagnostics 14, 2184 (2024).
-
de Almeida e Bueno, L., Kwong, M. T. & Bergmann, J. H. M. Performance of oral cavity sensors: a systematic review. Sensors 23, 588 (2023).
-
Li, Y., Ou, Y., Fan, K. & Liu, G. Salivary diagnostics: opportunities and challenges. Theranostics 14, 6969–6990 (2024).
-
Morales, M. A. & Halpern, J. M. Guide to selecting a biorecognition element for biosensors. Bioconjug. Chem. 29, 3231–3239 (2018).
-
Kim, J., Campbell, A. S., de Ávila, B. E.-F. & Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406 (2019).
-
Chambers, J. P., Arulanandam, B. P., Matta, L. L., Weis, A. & Valdes, J. J. Biosensor recognition elements. Curr. Issues Mol. Biol. 10, 1–12 (2008).
-
Luka, G. et al. Microfluidics integrated biosensors: a leading technology towards lab-on-a-chip and sensing applications. Sensors 15, 30011–30031 (2015).
-
Mummareddy, S., Pradhan, S., Narasimhan, A. & Natarajan, A. On demand biosensors for early diagnosis of cancer and immune checkpoints blockade therapy monitoring from liquid biopsy. Biosensors 11, 500 (2021).
-
Umapathy, V. R. et al. Emerging biosensors for oral cancer detection and diagnosis—a review unravelling their role in past and present advancements in the field of early diagnosis. Biosensors 12, 498 (2022).
-
Liu, Q. et al. Highly sensitive and wearable In2O3 nanoribbon transistor biosensors with integrated on-chip gate for glucose monitoring in body fluids. ACS Nano 12, 1170–1178 (2018).
-
Graf, H. & Mühlemann, H. R. Oral telemetry of fluoride ion activity. Arch. Oral Biol. 14, 259–IN3 (1969).
-
Gao, L., Tian, Y., Gao, W. & Xu, G. Recent developments and challenges in solid-contact ion-selective electrodes. Sensors 24, 4289 (2024).
-
Lee, Y. et al. Wireless, intraoral hybrid electronics for real-time quantification of sodium intake toward hypertension management. Proc. Natl. Acad. Sci. USA 115, 5377–5382 (2018).
-
Lim, H.-R. et al. Development of flexible ion-selective electrodes for saliva sodium detection. Sensors 21, 1642 (2021).
-
De Silva, T. et al. Ultrasensitive rapid cytokine sensors based on asymmetric geometry two-dimensional MoS2 diodes. Nat. Commun. 13, 7593 (2022).
-
Zargartalebi, H. et al. Active-reset protein sensors enable continuous in vivo monitoring of inflammation. Science 386, 1146–1153 (2024).
-
Hao, Z., Pan, Y., Shao, W., Lin, Q. & Zhao, X. Graphene-based fully integrated portable nanosensing system for on-line detection of cytokine biomarkers in saliva. Biosens. Bioelectron. 134, 16–23 (2019).
-
Chu, H. et al. A wearable electrochemical fabric for cytokine monitoring. Biosens. Bioelectron. 232, 115301 (2023).
-
Monajjemi, M. et al. A portable biosensor for simultaneous diagnosis of TNF-α and IL-1β in saliva biomarkers using twin electronic devices. J. Sens. Sens. Syst. 13, 245–261 (2024).
-
Bahri, M. et al. Capacitance electrochemical biosensor based on silicon nitride transducer for TNF-α cytokine detection in artificial human saliva: heart failure (HF). Talanta 209, 120501 (2020).
-
Zhakypbekova, A., Bekmurzayeva, A., Blanc, W. & Tosi, D. Parallel fiber-optic semi-distributed biosensor for detection of IL-6 and IL-8 cancer biomarkers in saliva at femtomolar limit. Opt. Laser Technol. 189, 113139 (2025).
-
Guerrero, S., Agüí, L., Yáñez-Sedeño, P. & Pingarrón, J. M. Design of electrochemical immunosensors using electro-click chemistry. Application to the detection of IL-1β cytokine in saliva. Bioelectrochemistry 133, 107484 (2020).
-
Yang, C.-Y. et al. Detection of picomolar levels of interleukin-8 in human saliva by SPR. Lab. Chip 5, 1017–1023 (2005).
-
Torrente-Rodríguez, R. M., Campuzano, S., Ruiz-Valdepeñas Montiel, V., Gamella, M. & Pingarrón, J. M. Electrochemical bioplatforms for the simultaneous determination of interleukin (IL)-8 mRNA and IL-8 protein oral cancer biomarkers in raw saliva. Biosens. Bioelectron. 77, 543–548 (2016).
-
Li, X. et al. A smartphone-based bacteria sensor for rapid and portable identification of forensic saliva sample. Sens. Actuators B Chem. 320, 128303 (2020).
-
Watkins, Z., Karajic, A., Young, T., White, R. & Heikenfeld, J. Week-long operation of electrochemical aptamer sensors: new insights into self-assembled monolayer degradation mechanisms and solutions for stability in serum at body temperature. ACS Sens 8, 1119–1131 (2023).
-
Archer, N., Ladan, S., Lancashire, H. T. & Petridis, H. Use of biosensors within the oral environment for systemic health monitoring—a systematic review. Oral 4, 148–162 (2024).
-
Batool, H. et al. Salivary levels of IL-6 and IL-17 could be an indicator of disease severity in patients with calculus associated chronic periodontitis. Biomed Res. Int. 2018, 8531961 (2018).
-
St. John, M. A. R. et al. Interleukin 6 and interleukin 8 as potential biomarkers for oral cavity and oropharyngeal squamous cell carcinoma. Arch. Otolaryngol. Neck Surg. 130, 929–935 (2004).
-
Mazurek-Mochol, M., Bonsmann, T., Mochol, M., Poniewierska-Baran, A. & Pawlik, A. The role of interleukin 6 in periodontitis and its complications. Int. J. Mol. Sci. 25, 2146 (2024).
-
Huang, Y.-K. et al. IL-8 as a potential therapeutic target for periodontitis and its inhibition by caffeic acid phenethyl ester in vitro. Int. J. Mol. Sci. 22, 3641 (2021).
-
Cheng, R., Wu, Z., Li, M., Shao, M. & Hu, T. Interleukin-1β is a potential therapeutic target for periodontitis: a narrative review. Int. J. Oral Sci. 12, 2 (2020).
-
Relvas, M. et al. Salivary IL-1β, IL-6, and IL-10 are key biomarkers of periodontitis severity. Int. J. Mol. Sci. 25, 8401 (2024).
-
Suryavanshi, V. G. et al. Assessment of IL-8 levels in saliva of healthy and chronic periodontitis individuals. J. Pharm. Bioallied Sci. 16, S825 (2024).
-
Márton, I. J. et al. Salivary IL-6 mRNA is a robust biomarker in oral squamous cell carcinoma. J. Clin. Med. 8, 1958 (2019).
-
Salim, A. et al. Salivary interleukin-6, interleukin-1β, and C-reactive protein as a diagnostic tool for plaque-induced gingivitis in children. Appl. Sci. 13, 5046 (2023).
-
Zhu, C., Zhao, Y. & Liu, J. Sensitive detection of biomarker in gingival crevicular fluid based on enhanced electrochemiluminescence by nanochannel-confined Co3O4 nanocatalyst. Biosensors 15, 63 (2025).
-
Bansal, T., Pandey, A., Deepa, D. & Asthana, A. K. C-reactive protein (CRP) and its association with periodontal disease: a brief review. J. Clin. Diagn. Res. JCDR 8, ZE21–ZE24 (2014).
-
Babaei, M., Rezaei, S., Saghafi Khadem, S., Shirinbak, I. & Basir Shabestari, S. The role of salivary C-reactive protein in systemic and oral disorders: a systematic review. Med. J. Islam. Repub. Iran 36, 138 (2022).
-
Mysak, J. et al. C-reactive protein in patients with aggressive periodontitis. J. Dent. Sci. 12, 368–374 (2017).
-
Radzki, D., Negri, A., Kusiak, A. & Obuchowski, M. Matrix metalloproteinases in the periodontium—vital in tissue turnover and unfortunate in periodontitis. Int. J. Mol. Sci. 25, 2763 (2024).
-
Checchi, V. et al. The role of matrix metalloproteinases in periodontal disease. Int. J. Environ. Res. Public. Health 17, 4923 (2020).
-
Sanikop, S., Patil, S. & Agrawal, P. Gingival crevicular fluid alkaline phosphatase as a potential diagnostic marker of periodontal disease. J. Indian Soc. Periodontol. 16, 513–518 (2012).
-
Domokos, Z. et al. Evaluating salivary MMP-8 as a biomarker for periodontal diseases: a systematic review and meta-analysis. Heliyon 10, e40402 (2024).
-
Arshad, R. et al. Salivary MMP-9 levels in chronic periodontitis patients with type-II diabetes mellitus. Molecules 27, 2174 (2022).
-
Rai, B., Kaur, J., Jain, R. & Anand, S. C. Levels of gingival crevicular metalloproteinases-8 and -9 in periodontitis. Saudi Dent. J. 22, 129–131 (2010).
-
Saleem, Z. et al. Estimation of salivary matrix metalloproteinases- 12 (MMP- 12) levels among patients presenting with oral submucous fibrosis and oral squamous cell carcinoma. BMC Oral Health 21, 205 (2021).
-
Monea, M. & Pop, A. M. The use of salivary levels of matrix metalloproteinases as an adjuvant method in the early diagnosis of oral squamous cell carcinoma: a narrative literature review. Curr. Issues Mol. Biol. 44, 6306–6322 (2022).
-
Hassan, S. H. S., El-Refai, M. I., Ghallab, N. A., Kasem, R. F. & Shaker, O. G. Effect of periodontal surgery on osteoprotegerin levels in gingival crevicular fluid, saliva, and gingival tissues of chronic periodontitis patients. Dis. Markers 2015, 341259 (2015).
-
Shojaee, M. et al. C – reactive protein levels in patients with periodontal disease and normal subjects. Int. J. Mol. Cell. Med. 2, 151–155 (2013).
-
Parsaie, Z., Rezaie, P., Azimi, N. & Mohammadi, N. Relationship between salivary alpha-amylase enzyme activity, anthropometric indices, dietary habits, and early childhood dental caries. Int. J. Dent. 2022, 2617197 (2022).
-
Macchia, E. et al. Selective single-molecule analytical detection of C-reactive protein in saliva with an organic transistor. Anal. Bioanal. Chem. 411, 4899–4908 (2019).
-
Reviansyah, F. H. et al. Noninvasive detection of alpha-amylase in saliva using screen-printed carbon electrodes: a promising biomarker for clinical oral diagnostics. Med. Devices Auckl. NZ 18, 15–27 (2025).
-
Sappia, L. et al. Electrochemical sensor for alkaline phosphatase as biomarker for clinical and in vitro applications. Sens. Actuators B Chem. 281, 221–228 (2019).
-
Mahato, K., Purohit, B., Kumar, A. & Chandra, P. Clinically comparable impedimetric immunosensor for serum alkaline phosphatase detection based on electrochemically engineered Au-nano-dendroids and graphene oxide nanocomposite. Biosens. Bioelectron. 148, 111815 (2020).
-
Shi, J.-J., He, T.-T., Jiang, F., Abdel-Halim, E. S. & Zhu, J.-J. Ultrasensitive multi-analyte electrochemical immunoassay based on GNR-modified heated screen-printed carbon electrodes and PS@PDA-metal labels for rapid detection of MMP-9 and IL-6. Biosens. Bioelectron. 55, 51–56 (2014).
-
Yang, G., Li, L., Rana, R. K. & Zhu, J.-J. Assembled gold nanoparticles on nitrogen-doped graphene for ultrasensitive electrochemical detection of matrix metalloproteinase-2. Carbon 61, 357–366 (2013).
-
Ren, X. et al. Increased electrocatalyzed performance through high content potassium doped graphene matrix and aptamer tri infinite amplification labels strategy: highly sensitive for matrix metalloproteinases-2 detection. Biosens. Bioelectron. 94, 694–700 (2017).
-
Chen, H. et al. Detection of matrilysin activity using polypeptide functionalized reduced graphene oxide field-effect transistor sensor. Anal. Chem. 88, 2994–2998 (2016).
-
Taylor, J. J. et al. A prototype antibody-based biosensor for measurement of salivary MMP-8 in periodontitis using surface acoustic wave technology. Sci. Rep. 9, 11034 (2019).
-
Tortolini, C. et al. A disposable immunosensor for the detection of salivary MMP-8 as biomarker of periodontitis. Bioelectrochemistry 156, 108590 (2024).
-
Nanda, J., Sachdev, V., Sandhu, M. & Deep-Singh-Nanda, K. Correlation between dental caries experience and mutans streptococci counts using saliva and plaque as microbial risk indicators in 3-8 year old children. A cross sectional study. J. Clin. Exp. Dent. 7, e114–e118 (2015).
-
Pannu, P., Gambhir, R. & Sujlana, A. Correlation between the salivary Streptococcus mutans levels and dental caries experience in adult population of chandigarh, india. Eur. J. Dent. 7, 191–195 (2013).
-
Lu, M., Xuan, S. & Wang, Z. Oral microbiota: a new view of body health. Food Sci. Hum. Wellness 8, 8–15 (2019).
-
Morrison, A. G., Sarkar, S., Umar, S., Lee, S. T. M. & Thomas, S. M. The contribution of the human oral microbiome to oral disease: a review. Microorganisms 11, 318 (2023).
-
Dinis, M. et al. Oral microbiome: Streptococcus mutans/caries concordant-discordant children. Front. Microbiol. 13, 782825 (2022).
-
Hemadi, A. S., Huang, R., Zhou, Y. & Zou, J. Salivary proteins and microbiota as biomarkers for early childhood caries risk assessment. Int. J. Oral Sci. 9, e1–e1 (2017).
-
Ge, Y., Caufield, P. W., Fisch, G. S. & Li, Y. Streptococcus mutans and streptococcus sanguinis colonization correlated with caries experience in children. Caries Res. 42, 444–448 (2008).
-
Kaman, W. E. et al. Highly specific protease-based approach for detection of Porphyromonas gingivalis in diagnosis of periodontitis. J. Clin. Microbiol. 50, 104–112 (2012).
-
Li, Y. et al. Portable saliva sensor based on dual recognition elements for detection of caries pathogenic bacteria. Anal. Chem. 96, 9780–9789 (2024).
-
Park, S., Park, K., Na, H. S., Chung, J. & Yang, H. Washing- and separation-free electrochemical detection of Porphyromonas gingivalis in saliva for initial diagnosis of periodontitis. Anal. Chem. 93, 5644–5650 (2021).
-
Witkowska, E., Łasica, A. M., Niciński, K., Potempa, J. & Kamińska, A. In search of spectroscopic signatures of periodontitis: a SERS-based magnetomicrofluidic sensor for detection of Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans. ACS Sens. 6, 1621–1635 (2021).
-
Dutta, G., Jallow, A. A., Paul, D. & Moschou, D. Label-free electrochemical detection of S. mutans exploiting commercially fabricated printed circuit board sensing electrodes. Micromachines 10, 575 (2019).
-
Shen, H. et al. A handheld electrochemical bacterial sensor based on multifunctional composite hydrogels and DNA biomimetic nanowalls for accurate detection of porphyromonas gingivalis. Chem. Eng. J. 498, 155344 (2024).
-
Kishen, A., John, M. S., Lim, C. S. & Asundi, A. A fiber optic biosensor (FOBS) to monitor mutans streptococci in human saliva. Biosens. Bioelectron. 18, 1371–1378 (2003).
-
Vasapollo, G. et al. Molecularly imprinted polymers: present and future prospective. Int. J. Mol. Sci. 12, 5908–5945 (2011).
-
Inonu, E., Hakki, S. S., Kayis, S. A. & Nielsen, F. H. The association between some macro and trace elements in saliva and periodontal status. Biol. Trace Elem. Res. 197, 35–42 (2020).
-
Romano, F. et al. Comparing ionic profile of gingival crevicular fluid and saliva as distinctive signature of severe periodontitis. Biomedicines 10, 687 (2022).
-
Chen, Z. et al. Hyperuricemia as a potential plausible risk factor for periodontitis. Med. Hypotheses 137, 109591 (2020).
-
Ye, L., Zhao, L., Mei, Z., Zhou, Y. & Yu, T. Association between periodontitis and uric acid levels in blood and oral fluids: a systematic review and meta-analysis. BMC Oral Health 23, 178 (2023).
-
Pampani, P. et al. Estimation of salivary sodium, potassium, and calcium levels in periodontal health and disease. J. Indian Soc. Periodontol. 28, 569–574 (2024).
-
Talwar, M., Tewari, A., Chawla, H. S. & Sachdev, V. A comparative assessment of fluoride concentration available in saliva using daily prescribed topical fluoride agents. Indian J. Dent. 7, 76–80 (2016).
-
Fejerskov, O., Thylstrup, A. & Larsen, M. J. Rational use of fluorides in caries prevention. A concept based on possible cariostatic mechanisms. Acta Odontol. Scand. 39, 241–249 (1981).
-
Buzalaf, M. A. R., Pessan, J. P., Honório, H. M. & Ten Cate, J. M. Mechanisms of action of fluoride for caries control. Monogr. Oral Sci. 22, 97–114 (2011).
-
Turtola, L. O. Salivary fluoride and calcium concentrations, and their relationship to the secretion of saliva and caries experience. Eur. J. Oral Sci. 85, 535–541 (1977).
-
Razzaque, M. S. Salivary phosphate as a biomarker for human diseases. FASEB Bioadvances 4, 102–108 (2022).
-
Samadi, F. M. et al. Quantitative correlation of serum and salivary trace elements in oral squamous cell carcinoma and oral potentially malignant disorders: an institution-based biochemical analysis. J. Oral Maxillofac. Pathol. JOMFP 28, 434–442 (2024).
-
Liu, Y., Li, M., Zhao, Z., Zhao, Z. & Li, M. A rapid and sensitive SERS-based Au/MIL-125(Ti) biosensor for hyperuricemia diagnosis via saliva analysis. Anal. Chem. 97, 6438–6446 (2025).
-
Bai, Y. et al. An inkjet-printed smartphone-supported electrochemical biosensor system for reagentless point-of-care analyte detection. Sens. Actuators B Chem. 346, 130447 (2021).
-
Kwan, R. C. H. et al. Amperometric biosensor for determining human salivary phosphate. Anal. Biochem. 343, 263–267 (2005).
-
Verhulst, M. J. L., Loos, B. G., Gerdes, V. E. A. & Teeuw, W. J. Evaluating all potential oral complications of diabetes mellitus. Front. Endocrinol. 10, 56 (2019).
-
Malon, R. S. P., Sadir, S., Balakrishnan, M. & Córcoles, E. P. Saliva-based biosensors: noninvasive monitoring tool for clinical diagnostics. Biomed Res. Int. 2014, 962903 (2014).
-
Gupta, S. et al. Correlation of salivary glucose level with blood glucose level in diabetes mellitus. J. Oral Maxillofac. Pathol. JOMFP 21, 334–339 (2017).
-
Schabmueller, C. G. J. et al. Micromachined sensor for lactate monitoring in saliva. Biosens. Bioelectron. 21, 1770–1776 (2006).
-
Nyein, H. Y. Y. et al. A wearable electrochemical platform for noninvasive simultaneous monitoring of Ca2+ and pH. ACS Nano 10, 7216–7224 (2016).
-
Farella, M. et al. Simultaneous wireless assessment of intra-oral pH and temperature. J. Dent. 51, 49–55 (2016).
-
Li, Y. et al. Oral wearable sensors: health management based on the oral cavity. Biosens. Bioelectron. X 10, 100135 (2022).
-
Lussi, A., Schlueter, N., Rakhmatullina, E. & Ganss, C. Dental erosion-an overview with emphasis on chemical and histopathological aspects. Caries Res. 45, 2–12 (2011).
-
Alzaid, M. et al. The effect of salivary pH on the flexural strength and surface properties of CAD/CAM denture base materials. Eur. J. Dent. 17, 234–241 (2023).
-
Li, Y., Mao, Y., Xiao, C., Xu, X. & Li, X. Flexible pH sensor based on a conductive PANI membrane for pH monitoring. RSC Adv 10, 21–28 (2019).
-
Srinivas, S., Ashokkumar, K., Sriraghavan, K. & Senthil Kumar, A. A prototype device of microliter volume voltammetric pH sensor based on carbazole–quinone redox-probe tethered MWCNT modified three-in-one screen-printed electrode. Sci. Rep. 11, 13905 (2021).
-
Brezniak, N. & Wasserstein, A. Root resorption following treatment with aligners. Angle Orthod 78, 1119–1124 (2008).
-
Ahmad, W., Xiong, J. & Xia, Z. Computational orthodontic force simulation: a review. Preprint at https://doi.org/10.48550/arXiv.2503.24195 (2025).
-
Rotolo, R. R., Aiyar, A., Perillo, L., Pedersen, T. K. & Stoustrup, P. Orthodontic and orthopedic management of dentofacial deformity from juvenile idiopathic arthritis: a systematic literature review. Eur. J. Orthod. 46, cjae023 (2024).
-
Grant, J. et al. Forces and moments generated by 3D direct printed clear aligners of varying labial and lingual thicknesses during lingual movement of maxillary central incisor: an in vitro study. Prog. Orthod. 24, 23 (2023).
-
Wang, S. et al. Wireless measurement of orthodontic forces in invisible aligners. Med. Nov. Technol. Devices 21, 100282 (2024).
-
Gold, B. P., Siva, S., Duraisamy, S., Idaayath, A. & Kannan, R. Properties of orthodontic clear aligner materials—a review. J. Evol. Med. Dent. Sci. 10, 3288–3294 (2021).
-
Maoddi, P. et al. Correlation between a force-sensing oral appliance and electromyography in the detection of tooth contact bruxism events. J. Clin. Med. 11, 5532 (2022).
-
Gu, J. et al. Self-powered strain sensor based on the piezo-transmittance of a mechanical metamaterial. Nano Energy 89, 106447 (2021).
-
Barlian, A. A., Park, W.-T., Mallon, J. R., Rastegar, A. J. & Pruitt, B. L. Review: semiconductor piezoresistance for microsystems. Proc. IEEE 97, 513–552 (2009).
-
Meng, Q., Lu, Y., Wang, J., Chen, D. & Chen, J. A piezoresistive pressure sensor with optimized positions and thickness of piezoresistors. Micromachines 12, 1095 (2021).
-
Liu, Y., Jiang, X., Yang, H., Qin, H. & Wang, W. Structural engineering in piezoresistive micropressure sensors: a focused review. Micromachines 14, 1507 (2023).
-
Lapatki, B. G., Bartholomeyczik, J., Ruther, P., Jonas, I. E. & Paul, O. Smart bracket for multi-dimensional force and moment measurement. J. Dent. Res. 86, 73–78 (2007).
-
Shi, Y. et al. An ultra-thin piezoresistive stress sensor for measurement of tooth orthodontic force in invisible aligners. IEEE Sens. J. 12, 1090–1097 (2012).
-
Liu, L., He, B., Zhuang, J., Zhang, L. & Lv, A. Force measurement system for Invisalign based on thin film single force sensor. Measurement 97, 1–7 (2017).
-
Martinelli, L., Hurschler, C. & Rosenbaum, D. Comparison of capacitive versus resistive joint contact stress sensors. Clin. Orthop. Relat. Res. 447, 214 (2006).
-
Hafner, J., Lapatki, B. G. & Paul, O. First Telemetric Smart Orthodontic Bracket for Therapeutic Applications. Sensors 1–4 (IEEE, 2018).
-
Sun, X. et al. A self-cleaning intraoral flex-occlusometer based on superhydrophobic capacitive sensors for dental health monitoring. J. Mater. Chem. A 12, 18423–18432 (2024).
-
Ogihara, T. et al. Development of a device for measuring bite force on individual teeth using a capacitive surface pressure distribution sensor. Pediatr. Dent. J. 35, 100348 (2025).
-
Ghosh, S., Qiao, W., Yang, Z., Orrego, S. & Neelakantan, P. Engineering dental tissues using biomaterials with piezoelectric effect: current progress and future perspectives. J. Funct. Biomater. 14, 8 (2023).
-
Testa, M. et al. A validation study of a new instrument for low cost bite force measurement. J. Electromyogr. Kinesiol. 30, 243–248 (2016).
-
Ramadan, K. S., Sameoto, D. & Evoy, S. A review of piezoelectric polymers as functional materials for electromechanical transducers. Smart Mater. Struct. 23, 33001 (2014).
-
Bani-Hani, M. & Amin Karami, M. Piezoelectric tooth aligner for accelerated orthodontic tooth movement. in 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 4265–4268 (IEEE, 2018).
-
Meenakshi, S. S. & Malaiappan, S. Piezosurgical corticotomy for rapid orthodontic tooth movement—a case series. J. Indian Soc. Periodontol. 25, 360–364 (2021).
-
Hu, J. et al. Flexible six-dimensional force sensor inspired by the tenon-and-mortise structure of ancient Chinese architecture for orthodontics. Nano Energy 96, 107073 (2022).
-
Feng, H. et al. A fully integrated orthodontic aligner with force sensing ability for machine learning-assisted diagnosis. Adv. Sci. 12, 2411187 (2025).
-
Perillo, L. et al. Monitoring biochemical and structural changes in human periodontal ligaments during orthodontic treatment by means of micro-Raman spectroscopy. Sensors 20, 497 (2020).
-
Torres-Rosas, R. et al. Force decay and elongation of orthodontic elastomeric chains exposed to different beverages common in the diet: an in vitro study. Dent. Med. Probl. 60, 413–420 (2023).
-
Albertini, P. et al. Stress relaxation properties of five orthodontic aligner materials: a 14-day In-vitro study. Bioengineering 9, 349 (2022).
-
Al-Nadawi, M. et al. Effect of clear aligner wear protocol on the efficacy of tooth movement. Angle Orthod. 91, 157–163 (2021).
-
Kuhl, M. et al. A telemetric stress-mapping CMOS chip with 24 FET-based stress sensors for smart orthodontic brackets. 2011 IEEE Int. Solid-State Circuits Conf. 108–110 (IEEE, 2011).
-
Kuhl, M. et al. A wireless stress mapping system for orthodontic brackets using CMOS integrated sensors. IEEE J. Solid-State Circuits 48, 2191–2202 (2013).
-
Gibbs, C. H., Mahan, P. E., Mauderli, A., Lundeen, H. C. & Walsh, E. K. Limits of human bite strength. J. Prosthet. Dent. 56, 226–229 (1986).
-
Pereira, L. J., Gavião, M. B. D., Bonjardim, L. R., Castelo, P. M. & van der Bilt, A. Muscle thickness, bite force, and craniofacial dimensions in adolescents with signs and symptoms of temporomandibular dysfunction. Eur. J. Orthod. 29, 72–78 (2007).
-
Williams, W. N., Low, S. B., Cooper, W. R. & Cornell, C. E. The effect of periodontal bone loss on bite force discrimination. J. Periodontol. 58, 236–239 (1987).
-
Kogawa, E. M., Calderon, P. S., Lauris, J. R. P., Araujo, C. R. P. & Conti, P. C. R. Evaluation of maximal bite force in temporomandibular disorders patients. J. Oral Rehabil. 33, 559–565 (2006).
-
Verma, T. P., Kumathalli, K. I., Jain, V. & Kumar, R. Bite force recording devices—a review. J. Clin. Diagn. Res. JCDR 11, ZE01–ZE05 (2017).
-
Braun, S. et al. A study of maximum bite force during growth and development. Angle Orthod. 66, 261–264 (1996).
-
Soni, R., Yadav, H., Pathak, A., Bhatnagar, A. & Kumar, V. Comparative evaluation of biting force and chewing efficiency of all-on-four treatment concept with other treatment modalities in completely edentulous individuals. J. Indian Prosthodont. Soc. 20, 312 (2020).
-
Serra, M. D., Gambareli, F. R. & Gavião, M. B. D. A 1-year intraindividual evaluation of maximum bite force in children wearing a removable partial dental prosthesis. J. Dent. Child. Chic. Ill 74, 171–176 (2007).
-
Slingsby, L. S., Jones, A. & Waterman-pearson, A. E. Use of a new finger-mounted device to compare mechanical nociceptive thresholds in cats given pethidine or no medication after castration. Res. Vet. Sci. 70, 243–246 (2001).
-
Bavia, P. F., Vilanova, L. S. R. & Garcia, R. C. M. R. Craniofacial morphology affects bite force in patients with painful temporomandibular disorders. Braz. Dent. J. 27, 619–624 (2016).
-
Assery Prosthodontics (Certified), M. K., Albusaily, H. S., Pani, S. C. & Aldossary, M. S. Bite force and occlusal patterns in the mixed dentition of children with Down syndrome. J. Prosthodont. 29, 472–478 (2020).
-
Emes, Y., Dergin, G. & Aybar, B. Temporomandibular Joint Pathology: Current Approaches and Understanding. (BoD – Books on Demand, 2018).
-
Nishigawa, K., Bando, E. & Nakano, M. Quantitative study of bite force during sleep associated bruxism. J. Oral Rehabil. 28, 485–491 (2001).
-
Chrcanovic, B. R. et al. Relationship between bite force, bruxism, and fractures of teeth and dental restorations. Sci. Rep. 15, 22752 (2025).
-
Kim, J. R. & Chung, J. W. The effect of stabilization splint use on bite force in patients with bruxism. J. Oral Med. Pain 49, 102–108 (2024).
-
Ichikawa, K. & Hijikata, W. Novel self-powered flexible thin bite force sensor with electret and dielectric elastomer. Sens. Mater. 34, 4237 (2022).
-
Flores-Ramírez, B., Suaste-Gómez, E., García-Limón, V. & Angeles-Medina, F. Flexible PVDF sensors for bruxism bite force measurement: a redefined instrumental approach. PLoS ONE 20, e0330422 (2025).
-
Noh, M. K. et al. Assessment of IL‑6, IL‑8 and TNF‑α levels in the gingival tissue of patients with periodontitis. Exp. Ther. Med. 6, 847–851 (2013).
-
Becerik, S. et al. Effects of menstrual cycle on periodontal health and gingival crevicular fluid markers. J. Periodontol. 81, 673–681 (2010).
-
Zhu, C., Wang, H. & Liu, J. Highly sensitive electrochemical immunosensor based on methylene blue-reduced graphene oxide nanocomposites as signal probes for IL-6 detection in gingival crevicular fluid samples. Front. Chem. 13, 1549927 (2025).
-
Pruna, R. et al. Novel nanostructured indium tin oxide electrode for electrochemical immunosensors: suitability for the detection of TNF-α. Electrochim. Acta 283, 1632–1639 (2018).
Acknowledgements
The authors acknowledge financial support from the National Institute of Biomedical Imaging and Bioengineering Award R21EB031455, National Science Foundation Biosensing Program Award 2339756, and University of Florida Research Opportunity Seed Grant.
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tai, Y., Li, Y., Mornay, K.M. et al. Biosensors in dental, oral and craniofacial applications. npj Biosensing 3, 14 (2026). https://doi.org/10.1038/s44328-026-00079-w
-
Received:
-
Accepted:
-
Published:
-
Version of record:
-
DOI: https://doi.org/10.1038/s44328-026-00079-w