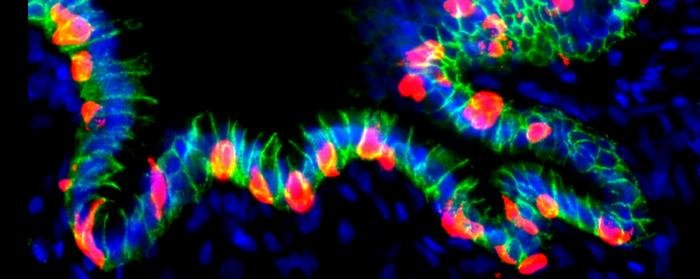
The explosion of mRNA vaccines brought on by the COVID-19 pandemic have proven that the delivery vehicles for those vaccines work incredibly well. Despite that, researchers are still working on improvements: to increase efficacy and reduce side effects from the vaccination. Now, researchers report modifications to lipid nanoparticles (LNPs) that outperform leading, commercially available formulations while reducing common vaccine side effects in preclinical tests of human cells and mouse models.
Changing the structure of the ionizable lipid of the LNPs boosted the metabolism of key immune cells, providing the energy necessary to gird the body’s defenses while dialing down the inflammatory signals that often cause fever and fatigue. The chemical tweak also enhanced on-target delivery of the nanoparticles to immune organs like the lymph nodes.
This work is published in Nature Materials in the paper, “Crosslinked ionizable lipids reprogram dendritic cell metabolism for potent mRNA vaccination.”
“This is an early step, but it opens the door to a new generation of mRNA vaccines that are more potent and better tolerated,” says Michael J. Mitchell, PhD, associate professor in bioengineering at the University of Pennsylvania. “Instead of accepting a trade-off between efficacy and side effects, we’re beginning to see that chemistry can help us improve both.”
Historically, LNPs have been viewed primarily as delivery vehicles. However, the data in this study demonstrate, for the first time, that LNPs also modify the metabolism of immune cells to enhance the efficacy of mRNA vaccines.

“We tweaked the standard lipid recipe by adding a new ingredient,” says Dongyoon Kim, PhD, a postdoctoral fellow in Mitchell’s lab, referring to imidoester cross-linkers, chemical groups whose connective ability expanded the possible range of ionizable lipid structures.
The best-performing lipid, C12-2aN, boosted the metabolism of dendritic cells. In both human dendritic cells and mouse models, LNPs built with C12-2aN increased the expression of genes involved in glycolysis and showed increased production of lactate without a loss in vaccine performance. In a mouse model of mRNA-based COVID-19 vaccination, the redesigned lipid performed on par with FDA-approved formulations. “This shows that we can make LNPs with more than one function,” says Kim. “With the right ingredients, these particles can deliver mRNA cargo and regulate immune cell metabolism at the same time.”
Normally, stronger immune activation within a vaccine comes with a cost. As immune cells ramp up to fight an active infection, they release inflammatory molecules that help coordinate the body’s response, but can also cause fever and fatigue. The new lipid appears to break that pattern.

“These lipids activate the immune system in a way that appears more controlled and confined to the immune cells,” says Amanda Murray, a doctoral student in the Mitchell Lab. “The dendritic cells are getting the energy they need to mount a protective vaccine response without triggering the same level of widespread inflammation that we normally experience after a vaccination, which causes common symptoms such as fever and muscle aches.”
Compared to an FDA-approved ionizable lipid, LNPs built with C12-2aN lowered the expression of genes associated with systemic inflammation in human cells and mice, and reduced levels of inflammatory markers in the mice’s bloodstreams. Mice that received the redesigned lipid also experienced smaller increases in body temperature than those treated with a standard lipid.
“Activating the immune system is essential for an effective vaccine,” adds Murray. “But thanks to the new lipid’s metabolic boost, that activation may be attainable without the usual side effects that make people complain about getting their vaccines.”
One challenge for LNPs is off-target delivery: nanoparticles accumulate preferentially in the liver instead of lymphoid organs. But with the C12-2aN lipid, the chemical changes also enhanced on-target delivery of LNPs. Compared to an FDA-approved formulation, the redesigned LNPs delivered more than three times as much mRNA to the lymph nodes relative to the liver.
The researchers also found that changes in lipid chemistry promoted glycolysis in other types of immune cells, suggesting that engineered ionizable lipids could help regulate immune cell metabolism in other diseases, including cancer, autoimmunity and other immune-mediated disorders.
“This study shows that rationally designing lipid chemistry allows us to do more than improve delivery,” says Mitchell. “We can begin to intentionally shape immune cell metabolism, opening new avenues for immune engineering beyond vaccines.”
The post Engineered LNPs Enhance mRNA Vaccine Efficacy, Reduce Side Effects appeared first on GEN – Genetic Engineering and Biotechnology News.