Data availability
The materials used in this study are available upon request. The scRNA-seq, omics data will be deposited under GEO accession numbers GSE267252 and GSE267067. The bulk RNA-seq, omics data will be deposited under GEO accession number GSE297701. Source data are provided with this paper.
References
-
Hagen, E. M. Acute complications of spinal cord injuries. World J. Orthop. 6, 17–23 (2015).
-
Sezer, N., Akkus, S. & Ugurlu, F. G. Chronic complications of spinal cord injury. World J. Orthop. 6, 24–33 (2015).
-
Ahuja, C. S. et al. Traumatic spinal cord injury. Nat. Rev. Dis. Primers 3, 17018 (2017).
-
Sofroniew, M. V. & Vinters, H. V. Astrocytes: biology and pathology. Acta Neuropathol. 119, 7–35 (2010).
-
Burda, J. E. & Sofroniew, M. V. Reactive gliosis and the multicellular response to CNS damage and disease. Neuron 81, 229–248 (2014).
-
Schnell, L., Fearn, S., Klassen, H., Schwab, M. E. & Perry, V. H. Acute inflammatory responses to mechanical lesions in the CNS: differences between brain and spinal cord. Eur. J. Neurosci. 11, 3648–3658 (1999).
-
Zhang, B. & Gensel, J. C. Is neuroinflammation in the injured spinal cord different than in the brain? Examining intrinsic differences between the brain and spinal cord. Exp. Neurol. 258, 112–120 (2014).
-
Alexander, J. K. & Popovich, P. G. Neuroinflammation in spinal cord injury: therapeutic targets for neuroprotection and regeneration. Prog. Brain Res. 175, 125–137 (2009).
-
David, S. & Kroner, A. Repertoire of microglial and macrophage responses after spinal cord injury. Nat. Rev. Neurosci. 12, 388–399 (2011).
-
Anderson, M. A., Ao, Y. & Sofroniew, M. V. Heterogeneity of reactive astrocytes. Neurosci. Lett. 565, 23–29 (2014).
-
Silver, J. & Miller, J. H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 5, 146–156 (2004).
-
Minev, I. R. et al. Biomaterials. Electronic dura mater for long-term multimodal neural interfaces. Science 347, 159–163 (2015).
-
Chen, M. S. et al. Nogo-A is a myelin-associated neurite outgrowth inhibitor and an antigen for monoclonal antibody IN-1. Nature 403, 434–439 (2000).
-
Lang, B. T. et al. Modulation of the proteoglycan receptor PTPsigma promotes recovery after spinal cord injury. Nature 518, 404–408 (2015).
-
Lu, P. et al. Long-distance growth and connectivity of neural stem cells after severe spinal cord injury. Cell 150, 1264–1273 (2012).
-
Tysseling-Mattiace, V. M. et al. Self-assembling nanofibers inhibit glial scar formation and promote axon elongation after spinal cord injury. J. Neurosci. 28, 3814–3823 (2008).
-
Alvarez, Z. et al. Bioactive scaffolds with enhanced supramolecular motion promote recovery from spinal cord injury. Science 374, 848–856 (2021).
-
Li, M. & Izpisua Belmonte, J. C. Organoids—preclinical models of human disease. N. Engl. J. Med. 380, 569–579 (2019).
-
Gupta, N. et al. Modeling injury and repair in kidney organoids reveals that homologous recombination governs tubular intrinsic repair. Sci. Transl. Med. 14, eabj4772 (2022).
-
Shoemaker, A. R. et al. Biofidelic dynamic compression of human cortical spheroids reproduces neurotrauma phenotypes. Dis. Models Mech. 14, dmm048916 (2021).
-
Henrique, D., Abranches, E., Verrier, L. & Storey, K. G. Neuromesodermal progenitors and the making of the spinal cord. Development 142, 2864–2875 (2015).
-
Gouti, M. et al. In vitro generation of neuromesodermal progenitors reveals distinct roles for wnt signalling in the specification of spinal cord and paraxial mesoderm identity. PLoS Biol. 12, e1001937 (2014).
-
Meinhardt, A. et al. 3D reconstitution of the patterned neural tube from embryonic stem cells. Stem Cell Rep. 3, 987–999 (2014).
-
Ogura, T., Sakaguchi, H., Miyamoto, S. & Takahashi, J. Three-dimensional induction of dorsal, intermediate and ventral spinal cord tissues from human pluripotent stem cells. Development 145, dev162214 (2018).
-
Gribaudo, S. et al. Self-organizing models of human trunk organogenesis recapitulate spinal cord and spine co-morphogenesis. Nat. Biotechnol. 42, 1243–1253 (2023).
-
Hendriks, D. et al. Human fetal brain self-organizes into long-term expanding organoids. Cell 187, 712–732.e738 (2024).
-
Andersen, J. et al. Generation of functional human 3D cortico-motor assembloids. Cell 183, 1913–1929.e1926 (2020).
-
Faustino Martins, J. M. et al. Self-organizing 3D human trunk neuromuscular organoids. Cell Stem Cell 26, 172–186.e176 (2020).
-
Xu, J. et al. Generation of neural organoids for spinal-cord regeneration via the direct reprogramming of human astrocytes. Nat. Biomed. Eng. 7, 253–269 (2023).
-
Zhang, Q. et al. Single-cell analysis reveals dynamic changes of neural cells in developing human spinal cord. EMBO Rep. 22, e52728 (2021).
-
Andersen, J. et al. Single-cell transcriptomic landscape of the developing human spinal cord. Nat. Neurosci. 26, 902–914 (2023).
-
Rayon, T., Maizels, R. J., Barrington, C. & Briscoe, J. Single-cell transcriptome profiling of the human developing spinal cord reveals a conserved genetic programme with human-specific features. Development 148, dev199711 (2021).
-
Escartin, C. et al. Reactive astrocyte nomenclature, definitions, and future directions. Nat. Neurosci. 24, 312–325 (2021).
-
Hartgerink, J. D., Beniash, E. & Stupp, S. I. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 294, 1684–1688 (2001).
-
Aida, T., Meijer, E. W. & Stupp, S. I. Functional supramolecular polymers. Science 335, 813–817 (2012).
-
Cui, H., Webber, M. J. & Stupp, S. I. Self-assembly of peptide amphiphiles: from molecules to nanostructures to biomaterials. Biopolymers 94, 1–18 (2010).
-
Beniash, E., Hartgerink, J. D., Storrie, H., Stendahl, J. C. & Stupp, S. I. Self-assembling peptide amphiphile nanofiber matrices for cell entrapment. Acta Biomater. 1, 387–397 (2005).
-
Silva, G. A. et al. Selective differentiation of neural progenitor cells by high-epitope density nanofibers. Science 303, 1352–1355 (2004).
-
Boekhoven, J. & Stupp, S. I. 25th anniversary article: supramolecular materials for regenerative medicine. Adv. Mater. 26, 1642–1659 (2014).
-
Alvarez, Z. et al. Artificial extracellular matrix scaffolds of mobile molecules enhance maturation of human stem cell-derived neurons. Cell Stem Cell 30, 219–238.e214 (2023).
-
Yuan, S. C. et al. Supramolecular motion enables chondrogenic bioactivity of a cyclic peptide mimetic of transforming growth factor-beta1. J. Am. Chem. Soc. 146, 21555–21567 (2024).
-
Edelbrock, A. N. et al. Supramolecular nanostructure activates TrkB receptor signaling of neuronal cells by mimicking brain-derived neurotrophic factor. Nano Lett. 18, 6237–6247 (2018).
-
Kozlowski, M. T., Crook, C. J. & Ku, H. T. Towards organoid culture without Matrigel. Commun. Biol. 4, 1387 (2021).
-
Berns, E. J. et al. Aligned neurite outgrowth and directed cell migration in self-assembled monodomain gels. Biomaterials 35, 185–195 (2014).
-
Brennan, F. H. et al. Microglia coordinate cellular interactions during spinal cord repair in mice. Nat. Commun. 13, 4096 (2022).
-
Rust, R. & Kaiser, J. Insights into the dual role of inflammation after spinal cord injury. J. Neurosci. 37, 4658–4660 (2017).
-
Liddelow, S. A. et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541, 481–487 (2017).
-
Park, D. S. et al. iPS-cell-derived microglia promote brain organoid maturation via cholesterol transfer. Nature 623, 397–405 (2023).
-
Schafer, S. T. et al. An in vivo neuroimmune organoid model to study human microglia phenotypes. Cell 186, 2111–2126.e2120 (2023).
-
Matson, K. J. E. et al. Single cell atlas of spinal cord injury in mice reveals a pro-regenerative signature in spinocerebellar neurons. Nat. Commun. 13, 5628 (2022).
-
Bellver-Landete, V. et al. Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat. Commun. 10, 518 (2019).
-
Bradbury, E. J. & Burnside, E. R. Moving beyond the glial scar for spinal cord repair. Nat. Commun. 10, 3879 (2019).
-
Rolls, A., Shechter, R. & Schwartz, M. The bright side of the glial scar in CNS repair. Nat. Rev. Neurosci. 10, 235–241 (2009).
-
Hellenbrand, D. J. et al. Inflammation after spinal cord injury: a review of the critical timeline of signaling cues and cellular infiltration. J. Neuroinflammation 18, 284 (2021).
-
Li, Y. et al. Microglia-organized scar-free spinal cord repair in neonatal mice. Nature 587, 613–618 (2020).
-
Lorach, H. et al. Walking naturally after spinal cord injury using a brain-spine interface. Nature 618, 126–133 (2023).
-
Boulting, G. L. et al. A functionally characterized test set of human induced pluripotent stem cells. Nat. Biotechnol. 29, 279–286 (2011).
-
Elmore, M. R. et al. Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 82, 380–397 (2014).
-
Haenseler, W. et al. A highly efficient human pluripotent stem cell microglia model displays a neuronal-co-culture-specific expression profile and inflammatory response. Stem Cell Rep. 8, 1727–1742 (2017).
-
Vaux, D. L., Fidler, F. & Cumming, G. Replicates and repeats—what is the difference and is it significant? A brief discussion of statistics and experimental design. EMBO Rep. 13, 291–296 (2012).
Acknowledgements
Research funding was provided by the Center for Regenerative Nanomedicine at Northwestern University and a gift from the John Potocsnak Family for spinal cord injury research. Peptide amphiphile synthesis was performed at the Peptide Synthesis Core Facility of the Center for Regenerative Nanomedicine at Northwestern University. The Peptide Synthesis Core is supported from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF ECCS-2025633). We are grateful to M. Karver, S. Biswas, E. Testa, J. Grzybek and T. Lusis for their assistance and advice on peptide synthesis. Analytical bioNanoTechnology Equipment Core (ANTEC) (RRID:SCR_023706) Facility of the Center for Regenerative Nanomedicine at Northwestern University is supported by the SHyNE Resource. We thank A. Kolot, H. Sai and K. Lee for general instrumentation service, and M. Seniw for the preparation of scientific illustrations at ANTEC. We thank X. Wang for service in bulk and single-cell RNA sequencing work supported by the Northwestern University NUSeq Core Facility. We thank C. Arvanitis, W. Liu and F. Korobova for the assistance in imaging work performed at the university core facility, Center for Advanced Microscopy generously supported by NCI CCSG P30 CA060553 awarded to the Robert H. Lurie Comprehensive Cancer Center. This work made use of the EPIC facility of Northwestern University’s NUANCE Center, which has received support from the SHyNE Resource (NSF ECCS-2025633), the IIN and Northwestern’s MRSEC programme (NSF DMR-2308691). We acknowledge the Robert H. Lurie Comprehensive Cancer Center of Northwestern University for the use of the Flow Cytometry Core Facility. The Lurie Cancer Center is supported in part by an NCI Cancer Center Support Grant number P30 CA060553. We appreciate G. Oliver (Northwestern University) for research instruments. We thank H. Sakaguchi (RIKEN), E. Kiskinis (Northwestern University), J. A. Ortega (University of Barcelona), Z. Alvarez (University of Barcelona) and J. Miska (Northwestern University) for technical advice and discussion. We would like to thank Northwestern undergraduate researchers, R. Gil, N. Coppisetti, T. Yang, J. McCubbin, N. Bruha and E. Lee for helping with stem cell organoid engineering projects.
Ethics declarations
Competing interests
N.T., N.A.S., Z.L. and S.I.S. are co-inventors on a patent application related to this work filed by Northwestern University (US patent application number 18/735,858, filed on 6 June 2024). The other authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Shi Yan Ng and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Inflammatory response after contusion injury of spinal cord organoids and addition of microglia.
(a) Schematic illustration of microglia assembly pathway 2. (b) Representative bright-field micrographs of mature microglia. (c) Schematic illustration of a protocol for the assembly 2. Mature microglia were assembled in freshly injured organoids (24-week) in the spinal cord maturation medium supplemented with IL-34 and mCSF. The organoids in 96 wells were then agitated at 55 rpm (revolutions per minute) using orbital shaking for 2 days before transder to 24 well dish. (d) Representative fluorescence micrographs of CSPG (green) and IBA-1 (magenta) and DAPI (blue). The square indicates high magnification image. (e) Schematic illustration showing the regions outlined by the red or black square, which indicates injured (inj.) or uninjured (uninj.) areas, respectively. Representative fluorescence micrographs of IBA1 (red) and GFAP (green). (f) Representative fluorescence micrographs of TUJ-1 (green) and DAPI (blue) in conditions with E2 PA or IKVAV PA2 (DAPI, blue). (g) Bar graphs of relative TUJ-1 intensity compared to uninjured area. Data are presented as mean values +/- s.e.m. of independently repeated experiments (n = 3; two-sided unpaired t-test). (h) Fluorescence micrographs of neurofilament (yellow) specific for axon represented in injured organoid surface treated with E2 PA or IKVAV PA2 (DAPI, blue). (i) Representative fluorescence micrographs of CSPG (red) treated with E2 PA or IKVAV PA2 (DAPI, blue). (j) Bar graph of relative CSPG intensity in injured versus uninjured areas. Data are presented as mean values +/- s.e.m. of independently repeated experiments (n = 3; two-sided unpaired t-test). Scale bars: 200 μm (d, f, h), 100 μm (b, e, i).
Supplementary information
Source data
Source Data Figs. 1–5, Extended Data Fig. 1
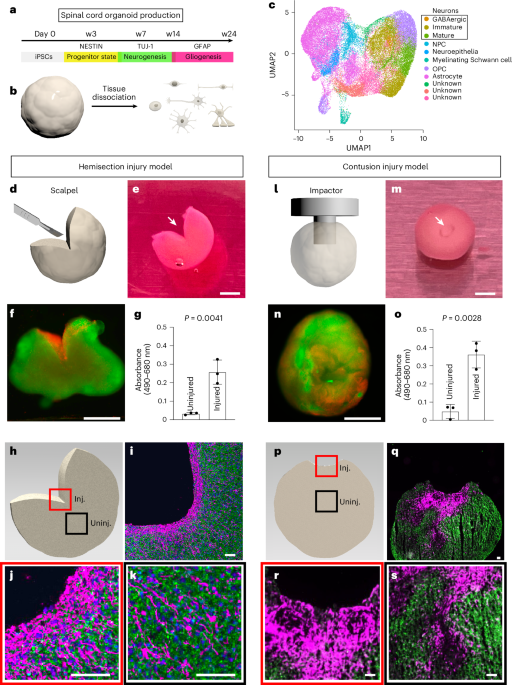
Figure 1. (g, o) Unprocessed absorbance intensities. Figure 2. (e) Intensities from fluorescence microscopy (‘w’ represents the condition after washing with culture medium); (o) average lengths of neurites measured by fluorescence microscopy; (p) average radial distribution of neurites relative to normal of organoid surface. Figure 3. (c, n) Average fluorescence intensities in injured regions relative to uninjured regions; (e, p) average lengths of neurites measured by fluorescence microscopy. Figure 4. (i) % of CD11b+/CD45+ cells measured by fluorescence-activated cell sorting; (j, k) normalized expression of C1QA and C3 relative to housekeeping genes; (m) average lengths of neurites measured by fluorescence microscopy; (q, s) average fluorescence intensities in injured regions relative to uninjured regions. Figure 5. Differential expression of genes detected by RNA sequencing. Extended Data Fig. 1. (g, j) Average fluorescence intensities in injured regions relative to uninjured regions.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takata, N., Li, Z., Metlushko, A. et al. Injury and therapy in a human spinal cord organoid. Nat. Biomed. Eng (2026). https://doi.org/10.1038/s41551-025-01606-2
-
Received:
-
Accepted:
-
Published:
-
Version of record:
-
DOI: https://doi.org/10.1038/s41551-025-01606-2