Introduction
The Hakamata case involved the murder of four family members of a managing director who worked at a miso processing factory in Shizuoka Prefecture in 1966, along with theft and arson. A factory employee, Iwao Hakamata, was arrested and denied the charges. However, his death penalty conviction was finalized in 1980 by the Supreme Court of Japan. The first request for a retrial was filed in 1981 but ended in 2008 when the Supreme Court dismissed a special appeal. In 2008, a second request for retrial was filed with the Shizuoka District Court, which in 2014 decided to start a retrial and suspended his execution and detention. Although the Tokyo High Court overturned this decision in 2018, the Supreme Court reinstated it in 2020, leading to further hearings. In March 2023, the Tokyo High Court supported the Shizuoka District Court’s retrial decision, and a retrial began in October 2023. On September 26, 2024, the defendant was declared not guilty after judges concluded that crucial evidence and the investigation records had been fabricated, with the prosecution choosing not to appeal (https://www.japantimes.co.jp/news/2024/09/26/japan/crime-legal/hakamata-retrial-ruling/).
Fourteen months after the murder, five pieces of blood-stained cotton clothes were found in a tank over a meter deep containing approximately eight tons of miso (fermented soybean paste). Testimonies and photographs taken at the time the clothes were discovered indicated that the blood stains were reddish in color. At the initial trial 45 years ago, the redness of the blood stains was not considered, and the clothing items were identified as those the defendant had been wearing at the time of the crime. As a result, the defendant was convicted. However, prior to the trial at Shizuoka District Court in 2014, the defense argued that blood stains would turn dark brown in a short period and would no longer remain red if placed in miso for over one year. Finally, in 2023, the Tokyo High Court decided to retry the case, considering it impossible for the blood stains to have remained red. The prosecutor asked one of the authors to prepare a scientific opinion on this issue and testify at the retrial, which began in 2023 (Shizuoka District Court, judgment date: September 26, 2024: https://www.courts.go.jp/app/hanrei_jp/search1).
Because it is challenging to determine whether a bloodstain remains red when placed in miso for > 1 year, we considered the formation of methemoglobin, which is generated through hemoglobin oxidation, and the subsequent degradation of hemoglobin as key events in the discoloration of blood stains1,2,3. Therefore, as a first step in our evaluation, we searched the literature on dissolved oxygen (DO) concentration in miso but found no relevant information. However, DO concentrations had been measured in other Japanese fermented foods, such as shoyu (soy sauce) and sake (Japanese rice wine)4,5,6.
Koji is a fermented product prepared by growing Aspergillus species in soybeans and/or grains such as rice and barley7,8. Miso is a traditional Japanese food made by mixing soybean pastes with rice koji, barley koji, soybean koji, and salt. It is then ripened for several months to over a year7,8. Aspergillus oryzae is most commonly used as a starter culture to prepare koji for making miso, and lactic acid bacteria and salt-tolerant yeasts, mainly Zygosaccharomyces rouxii, play important roles in miso maturation7,8. The koji mold in miso dies within a short time after preparation because of the high salt concentration (approximately ≥ 10%) in the mixture; however, enzymes present in koji, such as proteases, aminopeptidases, carboxypeptidases, L-glutaminases, amylases, and cellulases, remain functional over a long period7,8. These enzymes digest the starch and proteins found in grains and soybeans7,8. Although sake is fermented using rice koji made by growing A. oryzae, the type of yeast (Saccharomyces cerevisiae) used for fermentation differs from that used to make miso and shoyu9. Shoyu is produced by growing Aspergillus sojae or A. oryzae using only barley koji; however, the lactic acid bacteria and yeasts are very similar to those used in miso10. Previous studies indicated that the DO concentration in the supernatant of shoyu moromi, a soft solid formed during the fermentation process of multiple ingredients in the production of sake, shoyu, miso, and other fermented products, and a water-diluted sake moromi decreased to below the detection limit of the devices within a few days4,5,6. The detection limit was 0.1% O2 for the supernatant of shoyu moromi and 1 ppb for water-diluted sake moromi. However, in such studies, the DO concentrations in shoyu and sake were measured during brewing using the diaphragm electrode method4,5,6which is suitable for stirrable liquids but not for semisolid pastes such as miso11.
One report on miso suggested that the redox potential values were affected by DO12. Other reports indicated that the redox potential of miso was close to those of shoyu and sake13,14. The redox potential of miso is the sum of those of various redox substances, and the redox state of hemoglobin in miso is affected by the type and amount of these reducing substances as well as the DO concentration in miso. This value alone does not necessarily enable prediction of the redox state of hemoglobin. Additionally, if the DO level in miso is low, oxygenation of hemoglobin is reduced, decreasing the likelihood of hemoglobin auto-oxidation and subsequent formation of methemoglobin. Taken together, the findings of these studies indicate that, although measured indirectly, the DO concentration of miso is extremely low, similar to those of shoyu and sake during brewing. Seven forensic scientists, including one of the authors, prepared an expert report on this information before the retrial began, and one of the authors testified as such at the retrial.
Alternatively, the DO concentration can be measured using optical oxygen sensors, which were recently developed and mainly based on the principle of fluorescence quenching15. This method has been used to measure DO concentrations in the liquid and gas phases of wine, orange juice, and processed foods, as well as in strawberry puree and mayonnaise, which are semisolid pastes16,17,18,19,20,21,22. In the present study, we examined whether the DO concentration in miso could be measured using an optical oxygen sensor immediately after its preparation and in already-ripened miso.
Results
DO concentration in handmade Miso
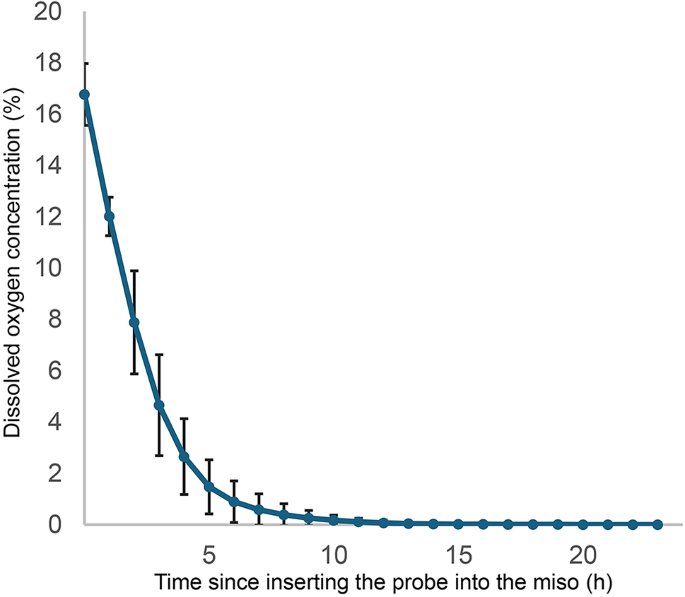
The DO concentration was measured at a depth of 5 cm in each of five laboratory-made miso samples (430 g) immediately after preparation. As shown in Fig. 1, the DO concentration at the time of preparation ranged from 14.8 to 17.9% (mean ± SD = 16.8 ± 1.2%); however, these initial values were outside the measurement range of the sensor (0–5% v/v O2). Values above and below the measurement range are displayed by this sensor but are not considered accurate. The device also indicated that the O2 concentration in the air was approximately 17.2–17.9%; however, based on a previous study, this value may have been approximately 20.9%23. Furthermore, the O2 concentration (or DO concentration) in 450 mL of distilled water placed in a 500-mL beaker and left in the atmosphere was almost the same as that in air, with a DO concentration of around 8 ppm. The actual DO concentration in miso at the time of preparation was considered to be slightly higher than these indicated values. Thereafter, the DO concentration gradually decreased and fell to below the detection limit (at 0.002% O2) between 9.5 and 23.2 h after preparation (mean ± SD = 14.8 ± 4.6 h, n = 5). As this method measures the partial pressure of oxygen at the tip of the probe, the value of the oxygen partial pressure of liquids placed in the atmosphere will be the same as that in air. Thus, the detection limit (an oxygen concentration of 0.002% or less) indicates that the oxygen partial pressure in that area is 0.015 mmHg or lower. Based on this information, our optical sensor can accurately measure DO concentrations of 0.002–5% O2 in miso. The DO concentration in miso appeared to decrease to < 0.002% within one day of preparation; however, the actual value and whether it was close to zero remained unclear. In addition, because the DO concentration decreases at the same oxygen partial pressure as salinity increases, the salinity of the homemade miso (12%) was incorporated into the software to correct the DO concentration values. At a salt concentration of 12%, the DO concentration was approximately 36% of the value without salt. After correcting for salinity, the DO concentration was less than 0.4 ppb, which was below the detection limit.
Dissolved oxygen (DO) concentration in miso immediately after preparation. DO measurements were taken every 3 min. The results of five experiments (mean ± SD) are displayed every hour.
Miso also contains high concentrations (15–20%) of sugars, including glucose24. Sugar and salt reduce the DO concentration25. Because the software used in this study did not allow for adjustments based on the sugar concentration, the actual DO concentration (ppb) is expected to be lower than the displayed value. Additionally, O2%, which is proportional to the oxygen partial pressure, is not affected by the salinity or sugar concentration. Therefore, in this study, the DO concentration was expressed in O2% rather than in ppb.
The DO concentration remained low for 2–36 h after falling below the detection limit. When the probe was removed and reinserted in a different location for measurement (from 1 to 19 days), the DO concentration fell to below the detection limit within 46 to 129 min (mean ± SD = 76.6 ± 30.9 min, n = 5). Hence, the DO concentration in the miso was still considered to be below the detection limit. Next, the miso was fermented in a plastic container at room temperature (controlled at 25 ± 1 °C) for approximately 1 month, then transferred to a glass beaker and fermented for 2 more months (total of 3 months). The DO concentration in the same miso was subsequently measured three times at 3-min intervals. The results showed that even for the same miso, the time required for the DO concentration to reach the detection limit varied between experiments, ranging from 24 to 132 min (mean ± SD = 81.0 ± 44.3 min, n = 3) (Fig. 2). Nonetheless, the DO concentration in miso could be measured using the probe and showed an extremely low value.
Dissolved oxygen (DO) concentration in miso approximately 3 months after preparation. DO concentration measurements were taken every 3 min, and the results of three experiments are displayed.
DO concentration in commercially available Miso
We measured the DO concentrations in five commercially available miso packages (Fig. 3). Miso A and B, made from rice koji, along with miso E, made from a combination of rice and barley koji, were raw miso samples containing live yeast. In contrast, miso C and D, made from rice koji, contained inactivated yeast. The DO concentration of each type of miso was measured twice. During the first measurement, the samples were left standing at room temperature until the packages were opened. All samples, except for Miso D, were packed with an oxygen absorber (small package containing particles primarily made of iron used to prevent the degradation of miso caused by oxidation resulting from oxygen trapped in the container). During the second measurement, the miso packages were stored in a refrigerator for 3–6 days and then returned to room temperature before the measurement. In the first round of measurements, 3.7–7.1 h were required for the DO concentration to fall below the detection limit, whereas during the second round, the DO concentrations, except for that of Miso E, took longer to fall below the detection limit (Table 1). Although the production date of these commercially available misos is unknown, we found that the DO concentration was extremely low, even in already-ripened miso. Furthermore, it was below the detection limit in raw miso containing live yeast (Fig. 3A, B, E) and in samples heated (or treated) with ethanol to inactivate the yeast (Fig. 3C, D). These results suggest that the oxygen consumed around the probe was not solely during yeast respiration. In addition, the DO concentration in miso was below the detection limit, regardless of oxygen absorber use or koji type.
Dissolved oxygen (DO) concentration evaluation in five commercially available miso samples. Miso A–D were rice koji miso, and miso E was a rice and barley koji miso.
Discussion
We measured the DO concentration in miso using an optical oxygen sensor to estimate whether the redness of bloodstains in miso would be maintained for more than a year. Previous reports suggested that the DO concentration in miso reaches nearly zero within 20–30 days, aligning with the redox potential stabilization timeline12. The redox potential change in miso is greatly affected by the DO concentration as well as the total amounts of redox substances, such as amino acids. Hence, it is difficult to accurately measure decreases in the DO concentration from the redox potential of miso. Notably, our findings obtained using an optical oxygen sensor revealed that the DO concentration in miso reached ≤ 0.002% in less than 1 day, which is much shorter than previous estimates. In addition, the DO concentration at a 5-cm depth in a 430-g sample may differ from that measured at a depth of 1 m or more in larger eight-ton tanks, where the blood-stained clothes were found. Although potentially lower in the latter situation, the DO concentration could not be verified, as it was beyond the detection limit of currently available oxygen sensors.
Conversely, during shoyu brewing, the DO concentration at a depth of 40 cm was estimated to be nearly zero, whereas at a depth of 10 cm, the oxygen absorption rate was 8% of that at the surface26. These findings are consistent with the fact that the amount of yeast per unit volume is much higher in shoyu moromi than in miso27. The DO concentration in sake is reportedly higher when brewed on a small scale than when brewed on a large scale28. This effect is attributable to differences in the viscosities of miso (130,000 mPa·s), a semisolid substance, and sake and shoyu (< 10 mPa·s), which are both liquids (https://tokisangyo.co.jp/wp-content/uploads/2022/01/syokuhin-data.pdf). Higher viscosity leads to minimal diffusion of oxygen (from the air on the surface) inside miso compared with that in shoyu or sake. Therefore, the DO concentration in miso is typically lower than those in shoyu and sake moromi, particularly near the surface, which contacts air.
By the time of the retrial, we had not measured the DO concentration in miso with an optical oxygen sensor; therefore, we could not conclusively testify that it was extremely low based on our findings. According to previous findings showing that yeast did not grow in miso except on the surface29,30one author testified that the DO concentration in miso, particularly near the bottom of large-scale tanks, may be extremely low, potentially close to zero. This opinion was rejected by the judges, who ruled that there was no scientific basis for extrapolating data obtained from studies involving shoyu to a semisolid like miso to prove that the DO concentration was that low.
The prosecutors subjected multiple samples to testing by preparing blood stains on cotton clothes and placing them in miso in plastic containers. Some of these containers contained oxygen absorbers, and some appeared to retain a reddish tinge after several months, regardless of the use of oxygen absorbers. Before the appeal trial at the Tokyo High Court in 2023, a defense witness submitted an expert report stating that the prosecutor’s experiments showed that the oxygen concentration in the miso was reduced to approximately 0.1% (absolute value of 0.8 mm Hg) of the atmospheric oxygen level by the oxygen absorber, which caused the blood stains to remain red for several months. This witness was later called to appear at the Tokyo High Court of Appeals in 2023 on remand to testify. He stated before the court that the oxygen concentration in sake is around 0.1% during brewing but is higher in miso, and this opinion was adopted. The Tokyo High Court finally ordered retrial of the case at the Shizuoka District Court. Before the retrial began, the prosecutor asked the defense witness, “What does it mean that the oxygen concentration in sake is around 0.1% during brewing?”, to which the witness replied in writing, “It is a relative concentration in the air, not an absolute concentration, which is 0.1% of the atmosphere, 0.02% in absolute concentration, which translates to approximately 5 ppb in DO concentration.” The defense witness, however, stated that the possibility of the DO concentration in sake during brewing being < 5 ppb could not be ruled out. Furthermore, when asked to provide evidence in support of the above statement by the prosecutor, the defense witness responded that they were referring to the reference in the expert report presented by the prosecution witnesses (including one of the authors) prior to the retrial5. However, the reference did not mention that the DO concentration in sake during brewing was approximately 5 ppb. Instead, it stated that it was below the detection limit of the instrument (1 ppb), and the tables in the reference indicated “<2 ppb”5. The court concluded, based on defense testimony, that even with a DO concentration as low as 5 ppb, oxygen persists in miso post-preparation, causing rapid heme oxidation during hemoglobin decomposition. The Shizuoka District Court in 2024 as well as the Tokyo High Court judges in 2023 concluded that the blood stains on cotton rags did not retain any redness after being placed in miso for > 1 year, aligning with the experimental results obtained by the aforementioned prosecutor. Thus, although redness is subjective and objectively determining whether blood stains retain redness over a given period is difficult, the Shizuoka District Court judges concluded that if the five pieces of clothing that the defendant was allegedly wearing at the time of the crime were placed in miso for 1 year and 2 months, the bloodstains would not retain any redness. Subsequently, they found the defendant not guilty because they determined that the clothes were not worn at the time of the crime and therefore could not have been placed there by the defendant. Thus, it was determined that the evidence was fabricated by investigators (Shizuoka District Court, judgment date: September 26, 2024: https://www.courts.go.jp/app/hanrei_jp/search1).
DO concentrations in shoyu and sake during brewing were measured in previous studies using the diaphragm electrode method4,5,6. However, direct measurement of the DO concentration in shoyu moromi was not possible because shoyu moromi is semisolid; hence, the supernatant of moromi was used6. In addition, sake moromi is not sufficiently liquefied until 5 days after brewing; therefore, a water-diluted moromi (dilution ratio of 2.2) was used for measurement5. These observations confirm that the diaphragm electrode method does not accurately measure the DO concentration of shoyu and sake immediately after brewing. In addition, the time required for the DO concentrations in both shoyu and sake immediately after brewing to reach values below the detection limit cannot be precisely determined using this method. Our results show that an optical oxygen sensor can measure the DO concentration even in semisolid pastes such as miso and is superior to the diaphragm electrode method, particularly when measuring the DO concentration in shoyu and sake moromi immediately after their preparation. Controlling the amount of oxygen in foods is necessary because it affects essential factors such as taste, nutritional value, color, and aroma. Based on our results, optical oxygen sensors can serve as effective tools for measuring the DO concentration in several foods, regardless of their viscosity. Given the strong impact of oxygen on food deterioration, employing this method to measure the oxygen concentration in food across different physical states (solid, semisolid, and liquid) will become increasingly important in the future.
Conclusion
Based on analysis of the DO concentrations of other fermented foods, such as sake and shoyu, as well as previous experiments on miso, the DO concentration of miso were predicted to be extremely low. Our measurement results of the DO concentration in miso align with this prediction.
Methods
Miso Preparation
Handmade miso was made using a miso kit (yield: 2 kg, consisting of 650 g of raw rice koji and 1.34 kg of mashed soybeans with salt) purchased from Suzuki Koji Store (Shizuoka, Japan). The raw materials were dispatched by courier; therefore, 3–6 days had already passed (since the koji was completed) by the time the miso was prepared. The composition of the miso in which the clothes were placed was unclear and could not be accurately reproduced using a commercial miso kit. However, we mixed the koji and mashed soybeans with salt in a proportion similar to that described in the written report. We prepared miso by combining 110 g of raw rice koji and 320 g of mashed soybeans with salt (final salinity of approximately 12%), mixed them well by hand, and removed as much air as possible. This mixture was placed in a 500-mL glass beaker at room temperature (controlled at 25 ± 1 ℃). The surface of the miso was covered with plastic wrap to retain moisture. The DO concentration was measured immediately and approximately 3 months after preparation (miso was fermented in a plastic container for approximately 1 month and then transferred to a glass beaker, where it was further fermented for another 2 months.
In addition, five types of commercially available miso (four rice koji miso, Fig. 3A–D, and one rice and barley koji miso, Fig. 3E) were purchased from a local supermarket, and their DO values were measured. Two rice koji miso (miso A: salinity 11.6%, 650 g and miso B: salinity 8.3%, 650 g) and one rice and barley koji miso (miso E: salinity 8.1%, 500 g) were raw miso containing live yeasts, whereas the other two rice koji miso (miso C: salinity 10.3%, 700 g and miso D: salinity 9.4%, 800 g) contained inactivated yeasts.
Oxygen measurements
The oxygen-sensing probe DP-PSt6 (PreSens-Precision Sensing GmbH, Regensburg Germany) with a measurement range of 0–5% v/v O2 or 0–2 mg dissolved O2/L was inserted at a depth of 5 cm into each of the miso samples (Fig. 4). According to the manufacturer’s instructions, DP-PSt6 consists of a polymer optical fiber, one end of which is coated with an oxygen-sensitive foil. The end of the fiber is covered with a high-grade steel tube to protect both the sensor material and the fiber. The steel tube has an outer diameter of 4 mm and a length of 10 cm. The DO concentration was measured every 3–10 min by connecting the probe to an OXY-1 SMA Trace, operated by PreSens Measurement Studio 2 software (PreSens-Precision Sensing GmbH)19,20,21,22. The OXY-1 SMA Trace device can be used for oxygen monitoring in several different sample types, including air, water, and sediment samples such as soil.
Handmade miso and the oxygen dipping probe DP-PSt6, OXY-1 SMA Trace for measuring dissolved oxygen (DO) concentration. The probe was inserted into the miso at a depth of approximately 5 cm.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- DO:
-
Dissolved oxygen
- koji :
-
Fermented product prepared by growing Aspergillus species
- miso :
-
Fermented soybean paste
- moromi :
-
Mash of multiple ingredients in the production of fermented products
- sake :
-
Japanese rice wine
- shoyu :
-
Soy sauce
References
-
Tsuruga, M., Matsuoka, A., Hachimori, A., Sugawara, Y. & Shikama, K. The molecular mechanism of autoxidation for human oxyhemoglobin. Tilting of the distal histidine causes nonequivalent oxidation in the beta chain. J. Biol. Chem. 273, 8607–8615 (1998).
-
Yasuda, J. et al. The alpha 1 beta 1 contact of human hemoglobin plays a key role in stabilizing the bound dioxygen. Eur. J. Biochem. 269, 202–211 (2002).
-
Shikama, K. & Matsuoka, A. Human haemoglobin: a new paradigm for oxygen binding involving two types of alphabeta contacts. Eur. J. Biochem. 270, 4041–4051 (2003).
-
Miyauchi, K., Inamori, K. & Uchida, K. Studies on environmental factors involved in fermentation of Shoyu Mash (part 2) effect of aeration on growth of Shoyu yeasts(1). J. Jpn Soy Sauce Res. Inst. 7, 13–18 (1981).
-
Nagai, H. Effect of dissolved oxygen concentration on sake brewing. J. Ferment. Technol. 70, 361–369 (1992).
-
Nagai, H. Effects of dissolved oxygen on sake brewing. J. Brew. Soc. Jpn. 88, 264–271 (1993).
-
Allwood, J. G., Wakeling, L. T. & Bean, D. C. Fermentation and the microbial community of Japanese Koji and miso: A review. J. Food Sci. 86, 2194–2207 (2021).
-
Kusumoto, K. I. et al. Japanese traditional Miso and Koji making. J. Fungi (Basel). 7, 579 (2021).
-
Kobayashi, T. et al. Modeling the sake brewing characteristics of rice from brown rice metabolites. J. Biosci. Bioeng. 134, 116–124 (2022).
-
Ito, K. & Matsuyama, A. Koji molds for Japanese soy sauce brewing: characteristics and key enzymes. J. Fungi (Basel). 7, 658 (2021).
-
Kinoshita, K. Preparation and properties of oxygen electrodes in Electrochemical Oxygen Technology (ed. Kinositah K.) 113–161 (Wiley, 1992).
-
Hondo, S. & Ysuhira, H. Relation between Oxidation-reduction potential and color of Miso. Sci. Technol. Miso. 43, 31–36 (1995).
-
Takeda, M. & Tukahara, T. Research on redox potential in sake brewing (part 1) regarding redox potential and turbidity. J. Brew. Soc. Jpn. 55, 51–54 (1960).
-
Mitsui, N. & Motokawa, E. Studies on the oxidation-reduction potentials of soy-sauce (part 1) on the oxido-reduction potentials of soy-sauce at the various stages of it’s production from riped Mash to commercial conditions. Jpn Soc. Biosci. Biotechnol. Agrochem. 41, 26–31 (1967).
-
Wang, X. D. & Wolfbeis, O. S. Optical methods for sensing and imaging oxygen: materials, spectroscopies and applications. Chem. Soc. Rev. 43, 3666–3761 (2014).
-
Kobayashi, H., Takamiya, M. & Hasegawa, M. Proterction against oxidation in mayonnaise by the control dissolved oxygen. Oleoscience 5 (3), 473–479 (2005).
-
Trivellin, N. et al. Study and development of a fluorescence based sensor system for monitoring oxygen in wine production: the WOW project. Sens. (Basel). 18, 1130 (2018).
-
Kim, A. N., Lee, K. Y., Han, C. Y., Kim, H. J. & Choi, S. G. Effect of an oxygen-free atmosphere during heating on anthocyanin, organic acid, and color of strawberry puree. Food Biosci. 50, 102065 (2022).
-
Rüegg, N., Röcker, B. & Yildirim, S. Application of palladium-based oxygen scavenger to extend the mould free shelf life of bakery products. Food Packag Shelf Life. 31, 100771 (2022).
-
Carrasco-Quiroz, M. et al. Influence of oxygen management on color and phenolics of red wines. Molecules 28, 459 (2023).
-
Coray, N. M. & Yildirim, S. Application of a Gallic acid–based oxygen scavenger label for the preservation of L-ascorbic acid in orange juice. Packag Technol. Sci. 37, 917–923 (2024).
-
Ureña, M. et al. Wine bottle overcapping wax: an aesthetic or functional element? Food Packag Shelf Life. 46, 101367 (2024).
-
Machta, L. & Hughes, E. Atmospheric oxygen in 1967 to 1970. Science 168, 1582–1584 (1970).
-
Hondo, S. Saccharides of Miso during manufacturing (Part 2). J. Brew. Soc. Jpn. 84, 594–599 (1989).
-
Sadler, G. D., Roberts, J. & Cornell, J. Determination of oxygen solubility in liquid foods using a dissolved oxygen electrode. J. Food Sci. 53, 1493–1496 (1988).
-
Miyauchi, K., Inamori, K. & Uchida, K. Studies on environmental factors involved in fermentation of Shoyu Mash (part 3) effect of aeration on growth of Shoyu yeasts (2). J. Jpn Soy Sauce Res. Inst. 7, 54–58 (1981).
-
Yoshii, H. Yeast (miso/soy sauce). J. Brew. Soc. Jpn. 67, 109–112 (1972).
-
Mtsuda, A. et al. Factors influencing sake quality, especially organic acid production, in a small scale brewery. J. Brew. Soc. Jpn. 108, 527–538 (2013).
-
Yoshii, H. Miso, soy sauce brewing and microorganisms. Chem. Biol. 8, 674–681 (1970).
-
Imai, S. & Matsumoto, I. Growing conditions of Torulopsis versatilis and Torulopsis etchellsii. J. Brew. Soc. Jpn. 70, 893–898 (1975).
Acknowledgements
We thank Dr. Hideaki Satou (Department of Medical Biochemistry, Kurume University School of Medicine) and Dr. Kwesi Teye (Kurume University Institute of Cutaneous Cell Biology) for their useful comments and discussions, and Ms. Katherine Ono and Editage (www.editage.com) for their assistance with the English language editing of this manuscript.
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Koda, Y., Soejima, M. Measuring dissolved oxygen in Miso for forensic medicine and semisolid food analysis. Sci Rep 15, 31666 (2025). https://doi.org/10.1038/s41598-025-17556-3
-
Received:
-
Accepted:
-
Published:
-
DOI: https://doi.org/10.1038/s41598-025-17556-3