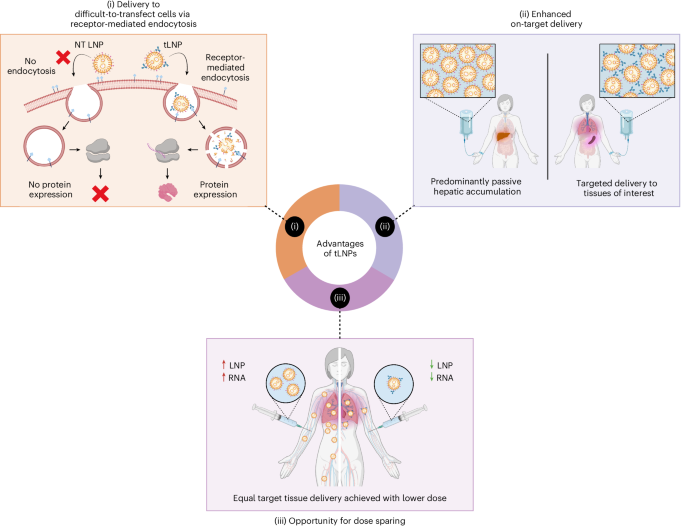
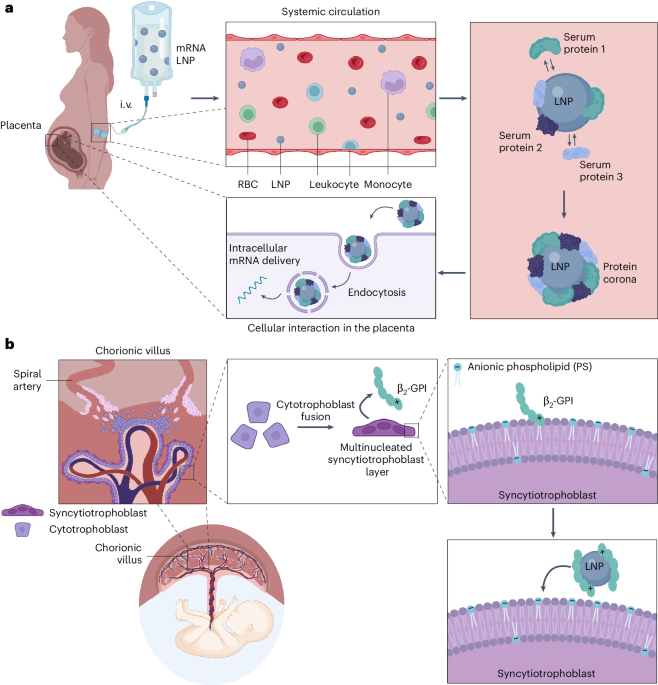
Hou, X., Zaks, T., Langer, R. & Dong, Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 6, 1078–1094 (2021).
Baden, L. R. et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 384, 403–416 (2021).
Polack, F. P. et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N. Engl. J. Med. 383, 2603–2615 (2020).
Cullis, P. R. & Felgner, P. L. The 60-year evolution of lipid nanoparticles for nucleic acid delivery. Nat. Rev. Drug Discov. 23, 709–722 (2024).
Swingle, K. L., Hamilton, A. G. & Mitchell, M. J. Lipid nanoparticle-mediated delivery of mRNA therapeutics and vaccines. Trends Mol. Med. 27, 616–617 (2021).
Wang, C., Zhang, Y. & Dong, Y. Lipid nanoparticle–mRNA formulations for therapeutic applications. Acc. Chem. Res. 54, 4283–4293 (2021).
Han, J. P. et al. In vivo delivery of CRISPR-Cas9 using lipid nanoparticles enables antithrombin gene editing for sustainable hemophilia A and B therapy. Sci. Adv. 8, eabj6901 (2022).
Rosenblum, D. et al. CRISPR-Cas9 genome editing using targeted lipid nanoparticles for cancer therapy. Sci. Adv. 6, eabc9450 (2020).
Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines—a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018).
Chaudhary, N., Weissman, D. & Whitehead, K. A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat. Rev. Drug Discov. 20, 817–838 (2021).
Akinc, A. et al. The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nat. Nanotechnol. 14, 1084–1087 (2019).
LoPresti, S. T., Arral, M. L., Chaudhary, N. & Whitehead, K. A. The replacement of helper lipids with charged alternatives in lipid nanoparticles facilitates targeted mRNA delivery to the spleen and lungs. J. Control. Release 345, 819–831 (2022).
Cheng, Q. et al. Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR–Cas gene editing. Nat. Nanotechnol. 15, 313–320 (2020).
Patel, S., Ryals, R. C., Weller, K. K., Pennesi, M. E. & Sahay, G. Lipid nanoparticles for delivery of messenger RNA to the back of the eye. J. Control. Release 303, 91–100 (2019).
Sago, C. D. et al. Nanoparticles that deliver RNA to bone marrow identified by in vivo directed evolution. J. Am. Chem. Soc. 140, 17095–17105 (2018).
Melamed, J. R. et al. Ionizable lipid nanoparticles deliver mRNA to pancreatic β cells via macrophage-mediated gene transfer. Sci. Adv. 9, eade1444 (2023).
Swingle, K. L. et al. Placenta-tropic VEGF mRNA lipid nanoparticles ameliorate murine pre-eclampsia. Nature 637, 412–421 (2025).
Chaudhary, N. et al. Lipid nanoparticle structure and delivery route during pregnancy dictate mRNA potency, immunogenicity, and maternal and fetal outcomes. Proc. Natl Acad. Sci. USA 121, e2307810121 (2024).
Billingsley, M. M. et al. Ionizable lipid nanoparticle-mediated mRNA delivery for human CAR T cell engineering. Nano Lett. 20, 1578–1589 (2020).
Ni, H. et al. Piperazine-derived lipid nanoparticles deliver mRNA to immune cells in vivo. Nat. Commun. 13, 4766 (2022).
Hosseini-Kharat, M., Bremmell, K. E. & Prestidge, C. A. Why do lipid nanoparticles target the liver? Understanding of biodistribution and liver-specific tropism. Mol. Ther. Methods Clin. Dev. 33, 101436 (2025).
Zhang, R. et al. Helper lipid structure influences protein adsorption and delivery of lipid nanoparticles to spleen and liver. Biomater. Sci. 9, 1449–1463 (2021).
Blanco, E., Shen, H. & Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 33, 941–951 (2015).
Dilliard, S. A., Cheng, Q. & Siegwart, D. J. On the mechanism of tissue-specific mRNA delivery by selective organ targeting nanoparticles. Proc. Natl Acad. Sci. USA 118, e2109256118 (2021).
Dilliard, S. A. & Siegwart, D. J. Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs. Nat. Rev. Mater. 8, 282–300 (2023).
Alavi, M. & Hamidi, M. Passive and active targeting in cancer therapy by liposomes and lipid nanoparticles. Drug Metab. Pers. Ther. 34, 20180032 (2019).
Menon, I., Zaroudi, M., Zhang, Y., Aisenbrey, E. & Hui, L. Fabrication of active targeting lipid nanoparticles: challenges and perspectives. Mater. Today Adv. 16, 100299 (2022).
Wang, Z. & Brenner, J. S. The nano-war against complement proteins. AAPS J. 23, 105 (2021).
Breda, L. et al. In vivo hematopoietic stem cell modification by mRNA delivery. Science 381, 436–443 (2023).
Shi, D., Toyonaga, S. & Anderson, D. G. In vivo RNA delivery to hematopoietic stem and progenitor cells via targeted lipid nanoparticles. Nano Lett. 23, 2938–2944 (2023).
Billingsley, M. M. et al. In vivo mRNA CAR T cell engineering via targeted ionizable lipid nanoparticles with extrahepatic tropism. Small 20, e2304378 (2024).
Metzloff, A. E. et al. Antigen presenting cell mimetic lipid nanoparticles for rapid mRNA CAR T cell cancer immunotherapy. Adv. Mater. 36, e2313226 (2024).
Veiga, N. et al. Cell specific delivery of modified mRNA expressing therapeutic proteins to leukocytes. Nat. Commun. 9, 4493 (2018).
Kedmi, R. et al. A modular platform for targeted RNAi therapeutics. Nat. Nanotechnol. 13, 214–219 (2018).
Marcos-Contreras, O. A. et al. Selective targeting of nanomedicine to inflamed cerebral vasculature to enhance the blood–brain barrier. Proc. Natl Acad. Sci. USA 117, 3405–3414 (2020).
Kim, Y. et al. Design of PD-L1-targeted lipid nanoparticles to turn on PTEN for efficient cancer therapy. Adv. Sci. 11, 2309917 (2024).
Tarab-Ravski, D. et al. Delivery of therapeutic RNA to the bone marrow in multiple myeloma using CD38-targeted lipid nanoparticles. Adv. Sci. 10, 2301377 (2023).
Parhiz, H. et al. PECAM-1 directed re-targeting of exogenous mRNA providing two orders of magnitude enhancement of vascular delivery and expression in lungs independent of apolipoprotein E-mediated uptake. J. Control. Release 291, 106–115 (2018).
Han, E. L. et al. Peptide-functionalized lipid nanoparticles for targeted systemic mRNA delivery to the brain. Nano Lett. 25, 800–810 (2025).
Carter, T., Mulholland, P. & Chester, K. Antibody-targeted nanoparticles for cancer treatment. Immunotherapy 8, 941–958 (2016).
Singh, R. et al. Dose-dependent therapeutic distinction between active and passive targeting revealed using transferrin-coated PGMA nanoparticles. Small 12, 351–359 (2016).
Nong, J. et al. Targeting lipid nanoparticles to the blood-brain barrier to ameliorate acute ischemic stroke. Mol. Ther. 32, 1344–1358 (2024).
Agard, N. J., Prescher, J. A. & Bertozzi, C. R. A strain-promoted [3 + 2] azide−alkyne cycloaddition for covalent modification of biomolecules in living systems. J. Am. Chem. Soc. 126, 15046–15047 (2004).
Han, X. et al. Ligand-tethered lipid nanoparticles for targeted RNA delivery to treat liver fibrosis. Nat. Commun. 14, 75 (2023).
Lin, Y., Cheng, Q. & Wei, T. Surface engineering of lipid nanoparticles: targeted nucleic acid delivery and beyond. Biophys. Rep. 9, 255–278 (2023).
Lee, D. et al. Strategies for targeted gene delivery using lipid nanoparticles and cell-derived nanovesicles. Nanoscale Adv. 5, 3834–3856 (2023).
Xue, L. et al. Rational design of bisphosphonate lipid-like materials for mRNA delivery to the bone microenvironment. J. Am. Chem. Soc. 144, 9926–9937 (2022).
Kasiewicz, L. N. et al. GalNAc-lipid nanoparticles enable non-LDLR dependent hepatic delivery of a CRISPR base editing therapy. Nat. Commun. 14, 2776 (2023).
Chen, D. et al. Rapid discovery of potent siRNA-containing lipid nanoparticles enabled by controlled microfluidic formulation. J. Am. Chem. Soc. 134, 6948–6951 (2012).
Shepherd, S. J., Issadore, D. & Mitchell, M. J. Microfluidic formulation of nanoparticles for biomedical applications. Biomaterials 274, 120826 (2021).
Shepherd, S. J. et al. Scalable mRNA and siRNA lipid nanoparticle production using a parallelized microfluidic device. Nano Lett. 21, 5671–5680 (2021).
Schellekens, H. Bioequivalence and the immunogenicity of biopharmaceuticals. Nat. Rev. Drug Discov. 1, 457–462 (2002).
Harding, F. A., Stickler, M. M., Razo, J. & DuBridge, R. The immunogenicity of humanized and fully human antibodies: residual immunogenicity resides in the CDR regions. mAbs 2, 256–265 (2010).
Pendley, C., Schantz, A. & Wagner, C. Immunogenicity of therapeutic monoclonal antibodies. Curr. Opin. Mol. Ther. 5, 172–179 (2003).
Safford, H. C. et al. Probing the role of lipid nanoparticle elasticity on mRNA delivery to the placenta. Nano Lett. 25, 4800–4808 (2025).
Yuan, Z., Yan, R., Fu, Z., Wu, T. & Ren, C. Impact of physicochemical properties on biological effects of lipid nanoparticles: are they completely safe. Sci. Total Environ. 927, 172240 (2024).
Hassett, K. J. et al. Impact of lipid nanoparticle size on mRNA vaccine immunogenicity. J. Control. Release 335, 237–246 (2021).
Muro, S. et al. Control of endothelial targeting and intracellular delivery of therapeutic enzymes by modulating the size and shape of ICAM-1-targeted carriers. Mol. Ther. 16, 1450–1458 (2008).
Swingle, K. L. et al. Ionizable lipid nanoparticles for in vivo mRNA delivery to the placenta during pregnancy. J. Am. Chem. Soc. 145, 4691–4706 (2023).
Geisler, H. C. et al. EGFR-targeted ionizable lipid nanoparticles enhance in vivo mRNA delivery to the placenta. J. Control. Release 371, 455–469 (2024).
Zamora, M. E. et al. Combination of physicochemical tropism and affinity moiety targeting of lipid nanoparticles enhances organ targeting. Nano Lett. 24, 4774–4784 (2024).
Zhao, G. et al. Precision treatment of viral pneumonia through macrophage-targeted lipid nanoparticle delivery. Proc. Natl Acad. Sci. USA 121, e2314747121 (2024).
Taiariol, L., Chaix, C., Farre, C. & Moreau, E. Click and bioorthogonal chemistry: the future of active targeting of nanoparticles for nanomedicines? Chem. Rev. 122, 340–384 (2022).
Dudchak, R. et al. Click chemistry in the synthesis of antibody-drug conjugates. Bioorg. Chem. 143, 106982 (2024).
Cabrera-Quiñones, N. C., López-Méndez, L. J., Cruz-Hernández, C. & Guadarrama, P. Click chemistry as an efficient toolbox for coupling sterically hindered molecular systems to obtain advanced materials for nanomedicine. Int. J. Mol. Sci. 26, 36 (2025).
Papp, T. E. et al. CD47 peptide-cloaked lipid nanoparticles promote cell-specific mRNA delivery. Mol. Ther. 33, 3195–3208 (2025).
Wang, Y., Wagner, C. R. & Distefano, M. D. Manipulating cell fates with protein conjugates. Bioconjug. Chem. 33, 1771–1784 (2022).
Martínez-Jothar, L. et al. Insights into maleimide-thiol conjugation chemistry: conditions for efficient surface functionalization of nanoparticles for receptor targeting. J. Control. Release 282, 101–109 (2018).
Marques, A. C., Costa, P. J., Velho, S. & Amaral, M. H. Functionalizing nanoparticles with cancer-targeting antibodies: a comparison of strategies. J. Control. Release 320, 180–200 (2020).
Fontaine, S. D., Reid, R., Robinson, L., Ashley, G. W. & Santi, D. V. Long-term stabilization of maleimide–thiol conjugates. Bioconjug. Chem. 26, 145–152 (2015).
Zhao, Z., Ukidve, A., Kim, J. & Mitragotri, S. Targeting strategies for tissue-specific drug delivery. Cell 181, 151–167 (2020).
Alibakhshi, A. et al. Targeted cancer therapy through antibody fragments-decorated nanomedicines. J. Control. Release 268, 323–334 (2017).
Bates, A. & Power, C. A. David vs. Goliath: the structure, function, and clinical prospects of antibody fragments. Antibodies 8, 28 (2019).
Richards, D. A., Maruani, A. & Chudasama, V. Antibody fragments as nanoparticle targeting ligands: a step in the right direction. Chem. Sci. 8, 63–77 (2017).
Richards, D. A. Exploring alternative antibody scaffolds: antibody fragments and antibody mimics for targeted drug delivery. Drug Discov. Today Technol. 30, 35–46 (2018).
Crivianu-Gaita, V. & Thompson, M. Aptamers, antibody scFv, and antibody Fab′ fragments: an overview and comparison of three of the most versatile biosensor biorecognition elements. Biosens.Bioelectron. 85, 32–45 (2016).
Asaadi, Y., Jouneghani, F. F., Janani, S. & Rahbarizadeh, F. A comprehensive comparison between camelid nanobodies and single chain variable fragments. Biomark. Res. 9, 87 (2021).
Jost, C. & Plückthun, A. Engineered proteins with desired specificity: DARPins, other alternative scaffolds and bispecific IgGs. Curr. Opin. Struct. Biol. 27, 102–112 (2014).
Stumpp, M. T., Binz, H. K. & Amstutz, P. DARPins: a new generation of protein therapeutics. Drug Discov. Today 13, 695–701 (2008).
Steeland, S., Vandenbroucke, R. E. & Libert, C. Nanobodies as therapeutics: big opportunities for small antibodies. Drug Discov. Today 21, 1076–1113 (2016).
Padilla, M. S. et al. Elucidating lipid nanoparticle properties and structure through biophysical analyses. Nat. Biotechnol. https://doi.org/10.1038/s41587-025-02855-x (2025).
Nogueira, S. S. et al. Analytical techniques for the characterization of nanoparticles for mRNA delivery. Eur. J. Pharm. Biopharm. 198, 114235 (2024).
Choi, Y. et al. Controlled spatial characteristics of ligands on nanoparticles: determinant of cellular functions. J. Control. Release 360, 672–686 (2023).
Yang, H., Le, Q.-V., Shim, G., Oh, Y.-K. & Shin, Y. K. Molecular engineering of antibodies for site-specific conjugation to lipid polydopamine hybrid nanoparticles. Acta Pharm. Sin. B 10, 2212–2226 (2020).
Lieser, R. M., Yur, D., Sullivan, M. O. & Chen, W. Site-specific bioconjugation approaches for enhanced delivery of protein therapeutics and protein drug carriers. Bioconjug. Chem. 31, 2272–2282 (2020).
Sapsford, K. E. et al. Functionalizing nanoparticles with biological molecules: developing chemistries that facilitate nanotechnology. Chem. Rev. 113, 1904–2074 (2013).
Cruz-Samperio, R. et al. Modular bioorthogonal lipid nanoparticle modification platforms for cardiac homing. J. Am. Chem. Soc. 145, 22659–22670 (2023).
Phillips, A. T. & Signs, M. W. Desalting, concentration, and buffer exchange by dialysis and ultrafiltration. Curr. Protoc. Protein Sci. 38, 4.4.1–4.4.15 (2004).
Clemmitt, R. H. & Chase, H. A. in Isolation and Purification of Proteins (eds Hatti-Kaul, R. & Mattiasson, B.) Ch. 10 (CRC Press, 2003).
Pohl, T. in Methods in Enzymology Vol. 182 (ed. Deutscher, M. P.) 68–83 (Academic Press, 1990).
Pieracci, J., Crivello, J. V. & Belfort, G. Photochemical modification of 10 kDa polyethersulfone ultrafiltration membranes for reduction of biofouling. J. Memb. Sci. 156, 223–240 (1999).
Guyon, L., Groo, A.-C. & Malzert-Fréon, A. Relevant physicochemical methods to functionalize, purify, and characterize surface-decorated lipid-based nanocarriers. Mol. Pharm. 18, 44–64 (2021).
Sapsford, K. E., Tyner, K. M., Dair, B. J., Deschamps, J. R. & Medintz, I. L. Analyzing nanomaterial bioconjugates: a review of current and emerging purification and characterization techniques. Anal. Chem. 83, 4453–4488 (2011).
Uddin, M. N. & Roni, M. A. Challenges of storage and stability of mRNA-based COVID-19 vaccines. Vaccines 9, 1033 (2021).
Zaleski, M. H. et al. Conjugation chemistry markedly impacts toxicity and biodistribution of targeted nanoparticles, mediated by complement activation. Adv. Mater. 37, 2409945 (2025).
Billingsley, M. M. et al. Orthogonal design of experiments for optimization of lipid nanoparticles for mRNA engineering of CAR T cells. Nano Lett. 22, 533–542 (2022).
Kauffman, K. J. et al. Efficacy and immunogenicity of unmodified and pseudouridine-modified mRNA delivered systemically with lipid nanoparticles in vivo. Biomaterials 109, 78–87 (2016).
Lee, Y., Jeong, M., Park, J., Jung, H. & Lee, H. Immunogenicity of lipid nanoparticles and its impact on the efficacy of mRNA vaccines and therapeutics. Exp. Mol. Med. 55, 2085–2096 (2023).
Korzun, T. et al. From bench to bedside: implications of lipid nanoparticle carrier reactogenicity for advancing nucleic acid therapeutics. Pharmaceuticals 16, 1088 (2023).
Thatte, A. S. et al. mRNA lipid nanoparticles for ex vivo engineering of immunosuppressive T cells for autoimmunity therapies. Nano Lett. 23, 10179–10188 (2023).
Tombácz, I. et al. Highly efficient CD4+ T cell targeting and genetic recombination using engineered CD4+ cell-homing mRNA-LNPs. Mol. Ther. 29, 3293–3304 (2021).
Ma, Y., VanKeulen-Miller, R. & Fenton, O. S. mRNA lipid nanoparticle formulation, characterization and evaluation. Nat. Protoc. 20, 2618–2651 (2025).
Wang, X. et al. Preparation of selective organ-targeting (SORT) lipid nanoparticles (LNPs) using multiple technical methods for tissue-specific mRNA delivery. Nat. Protoc. 18, 265–291 (2023).
Palanki, R. et al. Optimized microfluidic formulation and organic excipients for improved lipid nanoparticle mediated genome editing. Lab Chip 24, 3790–3801 (2024).
Wiener, J., Kokotek, D., Rosowski, S., Lickert, H. & Meier, M. Preparation of single- and double-oligonucleotide antibody conjugates and their application for protein analytics. Sci. Rep. 10, 1457 (2020).
Srinivasarao, M. & Low, P. S. Ligand-targeted drug delivery. Chem. Rev. 117, 12133–12164 (2017).
Mousavizadeh, A., Jabbari, A., Akrami, M. & Bardania, H. Cell targeting peptides as smart ligands for targeting of therapeutic or diagnostic agents: a systematic review. Colloids Surf. 158, 507–517 (2017).
Mariant, M., Camagna, M., Tarditi, L. & Seccamani, E. A new enzymatic method to obtain high-yield F(ab)2 suitable for clinical use from mouse IgGl. Mol. Immunol. 28, 69–77 (1991).
Lamoyi, E. & Nisonoff, A. Preparation of F(ab′)2 fragments from mouse IgG of various subclasses. J. Immunol. Methods 56, 235–243 (1983).
Crivianu-Gaita, V., Romaschin, A. & Thompson, M. High efficiency reduction capability for the formation of Fab’ antibody fragments from F(ab)2 units. Biochem. Biophys. Rep. 2, 23–28 (2015).
New England Biolabs. IdeZ Protease (IgG-specific). New England Biolabs https://www.neb.com/en-us/products/p0770-idez-protease-igg-specific (2026).
Hamilton, R. G. The Human IgG Subclasses (Pergamon Press, 1990).
Vidarsson, G., Dekkers, G. & Rispens, T. IgG subclasses and allotypes: from structure to effector functions. Front. Immunol. 5, 520 (2014).
Rousseaus, J., Biserte, G. & Bazin, H. The differential enzyme sensitivity of rat immunoglobulin G subclasses to papain and pepsin. Mol. Immunol. 17, 469–482 (1980).
Kinman, A. W. L. & Pompano, R. R. Optimization of enzymatic antibody fragmentation for yield, efficiency, and binding affinity. Bioconjug. Chem. 30, 800–807 (2019).
Nisonoff, A., Wissler, F. C., Lipman, L. N. & Woernley, D. L. Separation of univalent fragments from the bivalent rabbit antibody molecule by reduction of disulfide bonds. Arch. Biochem. Biophys. 89, 230–244 (1960).